2014 Group Project 4
| 2014 Student Projects | ||||
|---|---|---|---|---|
| 2014 Student Projects: Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | ||||
| The Group assessment for 2014 will be an online project on Fetal Development of a specific System.
This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Genital
Introduction
This page will explain the development of the genital system in the fetal stage of development, of both males and females, after briefly explaining the embryonic development. It explores the models of fetal development of the internal and external genitalia, including the timeline of development. This page also lists some animal models used in research and outlines some current research and findings regarding the development of the genital system, including historic findings. Finally, this page also discusses some of the congenital abnormalities of the genital system..
System Development
Prior to discussing the fetal development of the genital system, it is important to briefly appreciate the embryonic development of the genital tracts. Up until the seventh week of development, male and female genital tracts are indifferent. The genital system begins from a thickening of the epithelium surrounding the mesonephros, which lies dorsally in the coelomic cavity. The proliferation of this coelomic epithelium leads to an outgrowth known as the genital ridge. The genital ridge continues in outgrowth due to the ingression of the polemic epithelium, proliferation and recruitment of adjacent mesonephric cells. This bipotential genital ridge is indifferent in XX and XY embryos, however due to chromosomal and genetic influences differentiate into the testis and ovary respectively. [1]
Fetal genital development occurs in three main stages
1. Differentiation of gonad into Ovary or Testis
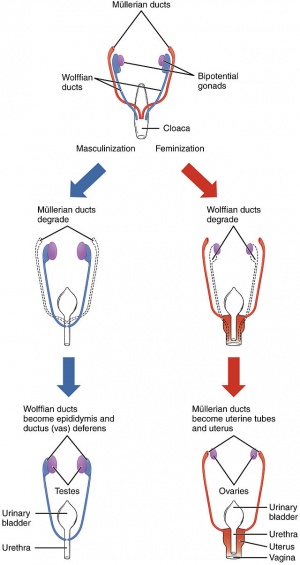
Internal Genital Development
| MALE | FEMALE |
| The fetal development of internal genitalia is largely dependent on the endocrine functions of the fetal testes. The fetal testes produce masculinizing hormones such as testosterone which begins its release from the interstitial Leydig cells of the primitive seminiferous tubules during the 8th week of development, and also the release of Mullerian Inhibiting Hormone (MIS) which is released at the sixth and seventh weeks by the Sertoli Cells. Testosterone acts primarily on the mesonephric ducts to stimulate the formation of the male genital ducts, whereas the MIS acts on the paramesonephric duct to stimulate its regression.
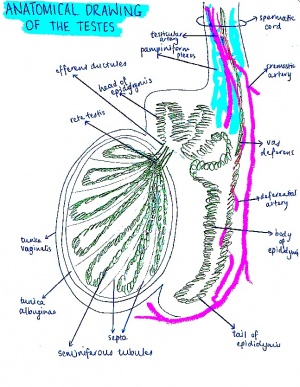
By the eighth week of fetal development in XY embryos, the testosterone produced in the testes results in the convolution of the proximal ends of the mesonephric ducts to form the 'epididymis. The mesonephros begins to degenerate, however some of the mesonephric tubules remain and develop into efferent ductules, which then open into the duct of the epididymis. Distal to this end, the mesonephric duct begins to develop a thick lining of smooth muscle and progresses to become the ductus deferens.
Fetal Male Glandular Development
|
The absence of testosterone production in XX embryos results in the regression of the mesonephric duct, and conversely the absence of MIH results in the development and progression of the paramesonephric ducts. The paramesonephric ducts give rise to most of the female internal genital system- the unfused cranial aspects of the ducts give rise to a primitive fallopian tubes, whilst the caudally fused portions form the uterovaginal primordium which will develop into a uterus and superior vagina. The splanchnic mesenchyme gives rise to the endometrial stromal tissue and the myometrium.
|
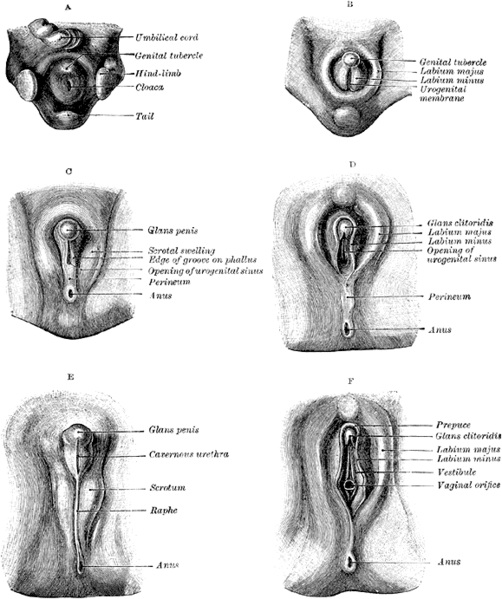
Development of the External Genitalia
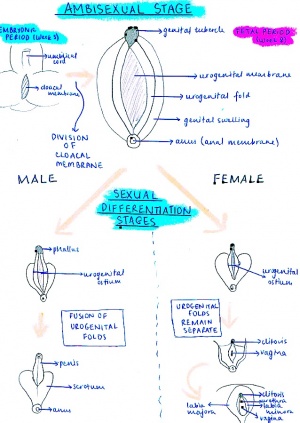
The current model for embryonic development of the external genitalia in humans and mice:
| Embryonic Period – fertilisation to end of 8th week (embryonic age) = AMBISEXUAL STAGE |
|---|
|
The current model for fetal development of the external genitalia in humans and mice:
| MALE MODEL | FEMALE MODEL |
Fetal Period – from 8th week of development = SEXUAL DIFFERENTIATION
|
Fetal Period – from 8th week of development = SEXUAL DIFFERENTIATION
|
Until the seventh week of development, the external genitalia of both XX and XY embryos is indistinguishable,. Sexual characteristics only become apparent after the ninth week, however are only clearly different between male and female after the twelfth week of development. In the fourth week of the embryonic phase of development, the cranial end of the cloacal membrane begins to swell and proliferate and produce a protrusion called the ‘”genital tubercle”’. This protrusion acts as a precursor for the major sexual organs, the glans penis in males and the glands clitoris in females.
Also in the forth week, swellings begin to form around the cloacal membrane called the labioscrotal swellings and urogenital folds. The genital tubercle elongates in both sexes and forms a primordial phallus. The urogenital membrane which lies on the floor of the cleft formed by the urethral folds
“’Male”’
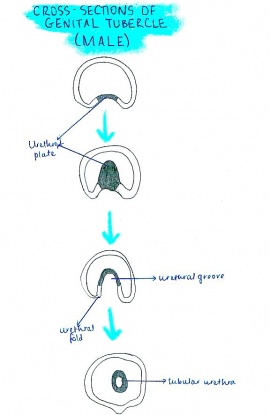
Testosterone produced by the interstitial Leydig cells of the fetal testes stimulates the masculinization of this indifferent genitalia. It stimulates the phallus to enlarges and elongates to form the “’glans penis”’, and the urethral folds begin to form the lateral walls of the urethral groove on the ventral aspect of the penis. A proliferation of endodermal cells lines the urethral groove and is referred to as the urethral plate, and it extends from the phallic portion of the urogenital sinus.
The urethral fold fuse with each other to form the ‘”spongy urethra”’ on the ventral surface of the penis. As the surface endoderm fuses in the median plane of the penis it encloses the spongy urethra- this fusion is called the “’penile raphe”’. At the tip of the penis, the an endodermal ingrowth forms a cellular endodermal chord which grows inside the penis towards the base. This cord canalizes and the lumen joins the previously formed spongy urethra, thus completing the terminal part of the urethra, mobbing the external urethral orifice to the tip of the penis.
During the twelfth week of development, an ectodermal circular ingrowth develops around the periphery of the penis and sits like a cap. The ingrowth breaks down and forms the ‘”prepuce”’ (foreskin) which remains as a fold of skin covering the tip of a penis. The mesenchyme of the phallus gives rise to the corpus cavernosum and the corpus spongiosum. The labioscrotal swellings grow toward eachother and fuse in the center to form the “’scrotum”’, this line of fusion is eferred to as the “’scrotal raphe”’.
“’Female”’ Much like the male genitalia, the genital tubercle swells and elongates to form the “’glans clitoris”’. At eighteen weeks, the clitoris is still a prominent swelling. In females, the urethral folds only fuse at the posterior aspect and form the ‘”labia minora’’. The labioscrotal folds fuse only at two points, posteriorly to form the posterior labial commissure, and anteriorly to form the “’mons pubis”’, the remain unfused portions remain as the “’labia majora”’.
References
[1] <pubmed>PMC3841730</pubmed> <pubmed>11315960</pubmed>
<pubmed>24240231</pubmed> <pubmed>24928207</pubmed> <pubmed>24741072</pubmed>
Related video
<html5media>https://www.youtube.com/watch?v=MureNA-RSZM</html5media>
Current Research, Models and Findings
Current Models
Most current research uses mouse models and observes the development of their external genitalia, especially their penile development, which initially appears to be different to human development. However, more microscopic inspection shows that mice have very similar external genitalia and are therefore appropriate animal models for observing such fetal development. As a result, mutant mouse models can effectively be used in future research to observe molecular mechanisms underlying hypospadias and their aetiology. [1]
When observing the morphology and cell biology of the developing testis, it is important to note that most of the research conducted on the subject involves the use of mouse models as a result of a lack of human subjects. It can be assumed that events in the human embryo correspond to the same events in the mouse embryo, however there are some differences between the time course of certain events and anatomy. [2]
Some examples of different animal models used in research involving fetal development of male and female genital systems are:
Different models are used for different research topics; for example, research involving Polycystic ovary syndrome (PCOS) uses murine models preferable due to the developmental time frame that allows studies of inherited PCOS to be examined within an appropriate time frame. The sheep model is also beneficial to use for PCOS research as it is cost effective and their size allows them to be subjected to certain procedures such as ultrasound and neurotransmitter measures. Primates are examples of an optimal model however are limited in their accessibility and long time frame of development.[7]
It is important to note that any findings associated with an animal model should be translated appropriately to the human model, as animals differ in their anatomy and regulatory mechanisms, as well as placentation. That is, sheep, rats and mice display different placentation and ovarian development that occurs in utero in sheep, primates and humans differs to the ex utero development in murine models.[7]
Current Research and Findings
Male
|
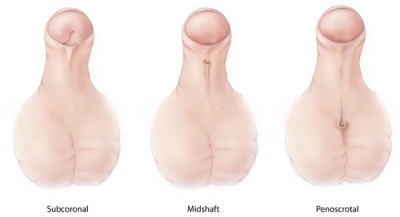
Extensive research into organogenesis of the external genitalia, mainly in males, is driven by the increasing incidence of hypospadias. Hypospadias are a result of the defect of fusion of the urethral folds of the lower part of the penis to fold and form the tubular penile urethra. The result of this in humans is the presence of an abnormal ventral urethral meatus, incomplete formation of the prepuce and an abnormal penile curvature.[1] Development of the male external genitalia, which occurs in the fetal period of development, is androgen dependent and involves epithelial-mesenchymal interactions. Because of these interactions, which are very similar to limb development, research into the development of genital tubercle has utilised similar methods for both processes. A minority of hypospadias cases are a result of the androgenic pathways being impaired and causing this congenital defect. The cell-cell interactions that allow for the development of the male external genitalia are mediated by a broad range of signaling molecules and growth factors such as fibroblast growth factors (FGFs), Sonic hedgehog (SHH) and bone morphogenetic proteins (BMPs). Such signaling and growth factors are downstream of androgen receptor signaling and an understanding of the mechanisms that underlie normal penile development during the fetal period, will lead to a deeper understanding of the aetiology of hypospadias.[1]
<pubmed>24030937</pubmed> The differentiation of the gonads into male or female begins around week 6-8 and is linked to the sex-determining region of the Y chromosome. The formation and descent of the testis is determined by a number of hormones which are:
The development of the male reproductive system requires the action of different hormones and is highly susceptible for development to be altered due to endocrine disruptions. Cryptorchidism is the failure of descent of the testis and is the most common congenital malformation in males. Non-steroidal anti-inflammatory drugs (NSAIDs) and paracetamol are some of the most widely used drugs used. These drugs have recently been identified as potential endocrine disruptors (ED) in humans. A number of epidemiological studies have reported that exposure to NSAIDs and analgesics during pregnancy showed an increased risk of cryptorchidism. This study used 62 fetuses from the first trimester between 7-12 weeks in gestation (GW) from pregnant women who obtained an abortion legally and with were given information and verbal consent was obtained according to national guidelines. The terminations were not motivated by abnormalities. The testes were cut in approximately 1mm3 pieces and drugs were used in the same concentration compared to recommended dosages in the body. The drugs used were paracetamol, aspirin, indomethacin, ketoconazole (antifungal). Testicular cells were counted using histology and image analysis and the hormones were assayed in the medium. The results showed no changes in the architecture of the testis with the analgesic treatment whereas the ketoconazole caused the boundaries of the testis cords to become unrecognisable. The analgesics did not significantly modify the number of germ cells or sertoli cells. Ketoconazole reduced testosterone levels in contrast to indomethacin, which stimulated testosterone production. Paracetamol had no significant effect on testosterone while aspirin produced a dose response relationship with an increase in testosterone after 72 hours in the youngest fetuses (8-9.86 GW) but not the older testes (10-12GW). None of the analgesics significantly affect the number of interstitial cells. The results showed a consistent trend for lower INSL3 production after 48-72 hours of exposure to mild analgesics and ketoconazole. This was the first study to measure direct production of INSL3 by the testis. Aspirin strongly stimulated AMH production, whereas as paracetamol and indomethacin increased production but not significantly. The analgesics did not significantly alter the sertoli cells and Ketoconazole significantly inhibited AMH production. Aspirin and paracetamol showed significant inhibition of Prostaglandin E2 production while indomethacin had no effect. In conclusion, the study shows that painkillers have a direct effect on various hormones, which are crucial for endocrine function and development of the human testis. The study shows that there is a direct effect with the dosages which are currently found with most medications. |
]
|