2012 Group Project 5: Difference between revisions
| Line 119: | Line 119: | ||
'''SOX-2''' | '''SOX-2''' | ||
The SOX-2 gene has been identified as the major causative gene of the two conditions. <ref> Verma,AS, FitzPatrick, DR,'''Anophthalmia and microphthalmia''' 2007 Orphanet Journal of Rare Diseases,pp.2-47 [http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/18039390]</ref>Although, SOX2 gene mutation is quite rare with an estimated frequency of 1 in 250,000 births, they are still considered the most common genetic cause of anophthalmia and microphthalmia to date. <ref> Slavotinek, AM, '''Eye development genes and known syndromes''' 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/22005280]</ref>Mutations in the SOX-2 gene is often associated with ocular malformations, they are variable in type but mostly bilateral and severe. This gene is highly expressed during lens induction process and the failure of this process may lead to anophthalmia. Also, the gene may have a role in the growth and maintenance of the developing lens. <ref>Ragge NK, Lorenz B, Schneider A, Bushby K, de Sanctis L et al '''SOX2 anophthalmia syndrome.''' 2005 Am J Med Genet A vol:135(1), pp.1-7[http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/15812812]</ref> | The SOX-2 gene has been identified as the major causative gene of the two conditions, it is located on the location 3q26.3-q27. It is an important transcription factor for maintaining self-renewal, or pluripotency, of undifferentiated embryonic stem cells. <ref> Verma,AS, FitzPatrick, DR,'''Anophthalmia and microphthalmia''' 2007 Orphanet Journal of Rare Diseases,pp.2-47 [http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/18039390]</ref>Although, SOX2 gene mutation is quite rare with an estimated frequency of 1 in 250,000 births, they are still considered the most common genetic cause of anophthalmia and microphthalmia to date. <ref> Slavotinek, AM, '''Eye development genes and known syndromes''' 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/22005280]</ref>Mutations in the SOX-2 gene is often associated with ocular malformations, they are variable in type but mostly bilateral and severe. This gene is highly expressed during lens induction process and the failure of this process may lead to anophthalmia. Also, the gene may have a role in the growth and maintenance of the developing lens. <ref>Ragge NK, Lorenz B, Schneider A, Bushby K, de Sanctis L et al '''SOX2 anophthalmia syndrome.''' 2005 Am J Med Genet A vol:135(1), pp.1-7[http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/15812812]</ref>SOX2 expression in humans are also seen in area of neural retina, optic stalk and lens suggesting its importance to the development of the eye.<ref> Slavotinek, AM, '''Eye development genes and known syndromes''' 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/22005280]</ref> | ||
SOX2 gene also co-operates with other genes to ensure normal lens development such as the [[PAX 6]] gene and they mutually induce each other.<ref> Slavotinek, AM, '''Eye development genes and known syndromes''' 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/22005280]</ref>It has been demonstrated in chicks, the co-regulation of these two gene drive lens induction in chicks, suggesting that lens induction failure in humans can be due to these genes. The lens induction is also mediated by the interaction of the two genes through their action on the γ-crystallin gene. <ref> Verma,AS, FitzPatrick, DR,'''Anophthalmia and microphthalmia''' 2007 Orphanet Journal of Rare Diseases,pp.2-47 [http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/18039390]</ref> | |||
'''PAX-6''' | '''PAX-6''' | ||
The PAX-6 gene mentioned before in the [[Abnormal lens development]] section has already been stated of its importance in lens development. The mutation of this gene cause alterantion to the developing lens and the pit of the optic vesicle.<ref> Slavotinek, AM, '''Eye development genes and known syndromes''' 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[http://www.ncbi.nlm.nih.gov.wwwproxy0.library.unsw.edu.au/pubmed/22005280]</ref> | |||
'''RAX''' | '''RAX''' | ||
Revision as of 00:08, 17 September 2012
Abnormal Vision
Introduction
The development of the eye occurs with the optic placode at a Carnegie stage 12 embryo (week 4). During this time any malfunction of development will create an abnormality of the eye. Abnormalities in vision can be acquired by environmental means, like that of Fetal Alcohol Syndrome or by Genetics such as the abnormalities discussed below. The purpose of this page is to give a brief overview of the development of the eye and some of the abnormalities which can occur.
Normal Eye Development
The normal early development of the eye begins with the optic primordium and sulcus developing in the neural folds at Carnegie stage 10 or around 22 days. Stage 11, or 24 days, sees the rostral neuropore closing and the optic vesicle forming from the optic sulcus. Only at stage 12 will you see the beginning formation of the optic placode as the optic vesicle sits near the surface ectoderm. [1] [2] [3] At stage 13, 28 days, a thickened surface ectoderm layer has developed on top of the optic vesicle, we know this to be the lens disc. The retinal disc (soon to be optic cup) has appeared on the wall of the optic vesicle.[4] As development continues through to Carnegie stage 14, the retinal disc is invaginated to form then optic cup and the lens pit forms. This lens pit closes in stage 15 and the optic cup and lens vesicle seem to bulge and press against the surface as the primary vitreous body begins to form.[5] [6] [7] This contour of the optic cup progresses until it is quite noticeable in stage 16, small grooves can also be seen above and below the eye. The retina differentiates at around stage 17 and the primary lens fibers obliterate the cavity of the lens vesicle filling the space at stage 18. The pupillary membrane and the layers of the cornea develop from stages 19-21. At stage 20 the retinal nerve fiber layer appears and grow towards the brain.[8] As the grooves above and below the eye develop and deepen in stage 17-19, eyelid folds develop, which soon turn into actual eyelids at around stage 19 and continue to develop further and grow slowly at stage 22. The eyelids close completely at stage 23.[9]
Abnormal Development
Abnormal Lens Development
As stated above, the lens originates from the ectoderm on top of the optic vesicle, it also requires a large portion of the head ectoderm surrounding this area before interacting with the optic vesicle. The vesicle is more involved with the correct positioning and lens formation at this stage. [10] [11] As the optic vesicle grows rapidly, the lens placode moves almost on top of the vesicle just close enough for a small gap to show. From this a network of fibrin adheres one surface to the other and the lens placode thickens and both sufaces start to invaginate becoming the lens pit and optic vesicle. The lens pit then detatches after deepening further and becomes the lens vesicle. From here the cells of the lens differentiate into primary fibres in the posterior half of the vesicle and epithelium in the anterior half. Rapid growth occurs with cell division occurring mostly in the epithelium region called the germinative zone with the daughter cells moving to the transitional zone where they mature and differentiate into fibre cells which continues throughout life. [12]
Pax-6 Genes
Most of the development of the lens occurs as a result of the Pax-6 gene. [13] [14] It is expressed in early developing optic vesicle and lens placode which is during the 4th and 5th week of human eye development. [15]It is responsible for the embryonic and postnatal development of the lens epithelium, and in particular, the ectoderm in embryonic development. And a variety of abnormalities can occur as a result of a mutation from this gene, Peters' anomaly (Corneal Opacity) and aniridia (lack of Iris) [16] are just some, as well as an abnormal cortical plate formations.[17] Within a few days of development, it is easy to distinguish an abnormality with the forebrain and the shape of the optic vesicle, as development continues there is an absence of a thickened ectodermal surface, lens placode, lens pit and the optic vesicle becomes distorted. As the ectodermal surface did not thicken, there is then no developing lens thus, Pax-6 gene is absolutely essential in the development of the lens. [18]
FOX genes
Another gene that may have an important role in the development of the lens is FOXe3 (Forkhead box protein E3) and FOXe4 (Forkhead box protein E4). They are expressed in the lens and they aid in the formation of the lens. Mutation of FOXe3 gene will cause anterior segment dysgenesis and result in cataracts formation as part of the dysgenesis. [19][20]FOXe3 can be detected in the first embryonic days 8.5, in two distinctive area of the cephallic neural folds. Both FOXe3 and FOXe4 are not as important as the Pax-6 gene, but it plays a very essential role in the control of proliferation and differentiation of the anterior len epithelium and it is evident that, the loss of these two genes will lead to anterior segment dysgenesis. [21]
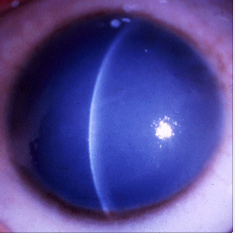
Congenital Cataracts (mutation of crystalline genes)
Cataracts of the eye can be defined as any opacity of the crystalline lens in which congenital cataract is especially important because it has the potential for inducing abnormal visual development and resulting in permanent blindness. Inherited cataracts contribute the most to congenital cataracts especially in developed countries. [22][23] Crystalline genes in the human body codes for major structural proteins in the lens, they are considered important due to their high level of expression in the lens and their functions in maintaining lens transparency. There is a strong relationship between their mutation and the development of congenital cataracts. [24] One example of crystalline gene is βγ-crystallins. When this gene is mutated, it causes major abnormalities in the protein structure and result in an unstable protein that can precipitates from solution and cause protein denaturation and precipitation which eventually leads to cataract formation.[25]Mutations in the αA-crystallin gene have also been implicated both in autosomal recessive and autosomal dominant cataract. One would expect that αB-crystallin would have a similar effect on cataracts formation but experiments suggested that cataracts formation is dominated by mutations in αA-crystallin gene with the abnormal protein having a toxic effect on lens cells. The mutant protein also inhibit the functioning of normal αA-crystallin protein. [26]
Images of congenital hereditary cataracts from mutations of crystallin genes
Abnormal Corneal Development
The corneal Development begins at around Carnegie Stage 15 with the surface ectoderm differentiating into the anterior epithelieum of the cornea, at this stage it has its own basement membrane. At stage 18 the posterior epithelium of the cornea begins to form and by stage 19, is easily recogniseable. [27] Stage 20 sees the developing cornea with an anterior epithelium, a postepithelial layer, and a posterior epithelium. The postepithelial layer will develop further to become the substantia propria of the cornea in stage 21 via cells invading the layer. This process is finished in stage 22 and at stage 2 the cornea consists of an anterior epithelium and a basement membrane, the substntia propria and the posterior epithelium. [28]
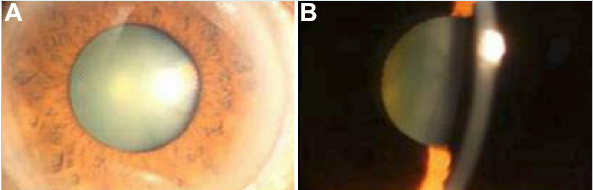
SCL4A11 gene
Congenital Autosomal recessive corneal endothelial dystrophy type 2(CHED2) is associated with mutations in SLC4A11, Solute Carrier family 4 (sodium borate cotransporter) member 11). This gene is located on chromosome 20p13-12. Mutation of this gene cause disorder of the cornea that is characterised by diffuse bilateral corneal clouding and they are often edematous and have a ground glass appearance that is evident at birth or in the neotal period and can cause the impairment of vision and require corneal transplantation.[29][30][31]
SLC4A11 is an electrogenic Na/borate cotransporter and it can stimulate cell growth and proliferation by increasing intracellular borate and activating the MAPK pathway.[32] The ion transporter SLC4A11 promote sodium-dependent transport of borate as well as flux of sodium and hydroxyl ions in vitro. It has been shown that SLC4A11 is expressed in the endothelial cells of the cornea where severe morphological changes of the cornea can be caused by SLC4A11 increasing the sodium chloride concentrations in the stroma.[33]SLC4A11 may also have a role in the growth and terminal differentiation of neural crest cells during the formation of the endothelium, the functional loss of the gene causes the death of endothelial cells and loss of barrier function, eventually secondary corneal edema. [34] Some other studies have shown that borate can lead to the phosphorylation of both MAP-kinase and extracellular signalling kinases, and these are part of the mitogen activated protein kinase cascade (MAPK). MAPKs are important in the regulation of cell cycle and growth, a deregulated MAPK pathway can lead to some of the morphological features in CHED2. [35]
Appearance of cornea due to CHED
Abnormal Retinal Development
The retina is of neuro-ectodermal origin[36] and is derived from the optic cup (stage 17). The optic cup is divided into the neural retina and the retinal pigmental epithelium. The neural retina is light sensitive, the region from which the photoreceptors and cell bodies of the neurons are located. As the retina is the part of the eye from which the brain receives the most visual information there are many abnormalities which can occur here during development. In various research avenues for Leber Congenital Amaurosis (to be discussed later) the following genes have been found to be involved in retinal development, they are as follows : RPE65 for retinoid metabolism, GUCY2D phototransduction, CRX photoreceptor outer segment development, RPGRIP1 disk morphogenesis, CRB1 zonula adherens formation and AIPL1 cell-cycle progression.[37]
CRX gene
An example of one of these gene expressions is CRX. CRX is expressed abundantly in photoreceptor cells and is an important regulator of various photoreceptor specific genes and key enzymes for melanin synthesis[38]. While it has been determined that CRX is important in terminal differentiation of the photoreceptors, using a mouse model, in which the otx2 was conditionally knocked out there was a complete loss of retinal photoreceptors and thus it has been found that otx2 is essential for CRX transcription as it is an upstream regulator of CRX expression[39]. Otx2 expression covers most of the fore and midbrain neuro-epithelium and subsequently the retinal region[40].
RPE65
RPE65 is an enzyme located in the retinal pigment epithelium (RPE)which is a catalyst during a crutial part of the visual cycle. It permits photoreceptors pigments to absorb photons which maintain sight[41].
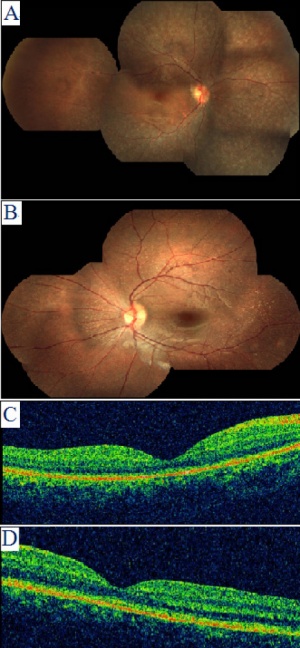
Retinal Pigment Epithelium and Albinism
Some research suggests that Retinal Pigment Epithelium (RPE) has a regulatory effect upon neural retina development[42]. A melanin related agent, has been suggested to be important in retinal development and maintenance during maturity as in albinism there is a reduction in melanin and retinal abnormalities are present[43]. The Tyrosinase gene controls melanin production and acts a catalyst in DOPA production from tyrosine and is a regulator of cell cycles[44]. Although there is evidence for a relationship between DOPA and retinal mitosis the mechanisms responsible are not so easily identifiable[45]. When melanin is absent a variety of retinal disorders such as abnormal connection between the eye and brain, undeveloped central retina and rod defects can occur, this reduction and/or absence of melanin is commonly caused by Albinism which also has reduced cell density is also abnormally low with ganglion cells of the retina decreased by 25%[46]. Other eye defects caused by Albinism are absent Fovea, undeveloped Macula region and abnormal chiasmatic projections to name a few[47].
Comparison between fundus in Albinism and Normal eye
Ocular Manifestations
Major ocular disorders can be split into two separate sections based on the way in which they originated.
Genetic
Leber Congenital Amaurosis
Leber Congenital Amaurosis (LCA) is an inherited retinal degenerative disorder that causes blindness or loss of sight at birth.[48] LCA has a recessive pattern of inheritance where there is a 25% chance for a child to contract LCA.[48]
LCA was first recognized by Dr Theodor Leber in 1869. He published the paper Ueber Retinitis pigmentosa und angeborene Amaurosa (About Retinitis pigmentosa and congenital Amaurosa) in Archiv fur Ophthalmologie (now known as Graefes Archive of clinical and experimental ophthalmology).[49] In his research Leber describes a set of clinical manifestations in children he studied at the Ilvesheim School for the blind in Germany with Retinitis Pigmentosia which are used still today as a diagnostic technique for LCA [50]. These manifestations are: sever visual loss at/near birth, wandering nystagmus, amaurotic pupils, and pigmentary retinopathy[50]. In his paper Leber classified this disease as being a part of the Retinitis Pigmentosia group of optical disorder, placing great emphasis upon the high incidence of hereditary factors[50]. As stated in the section on Abnormal Retinal Development out of the eighteen genes [51] found that contribute to LCA, six of them have been linked to specific sections to retinal development[50]. As a result making a differential diagnosis on congential blindness can be difficult as LCA can overlap with many other disorders such as Bardet-Biedl syndrome and Senior-Loken syndrome for example[51]. An important diagnostic technique was recognised by Franeschetti and Dieterle of non-detectable or severely reduced electroretinogram (ERG) measured in early progression of LCA[50]. The measurement of ERG has become a prerequisite diagnostic protocol for LCA[50].
CEP290 gene mutation is the most common cause of LCA and accounts for 6-22% of all cases[51]. This mutation is a slow progressive form of LCA and has a relatively normal optic disc with sclera ring. Abnormalities associated with CEP290 mutation include salt and pepper aspect of fundus with Macular degeneration causes typical Retinitis Pigmentosa appearance in second decade and Mild lobar retinal pigment epithelium atrophy[51]. Clinical signs associated with CEP290 include sluggish pupillary reflexes and juvenile cataracts[51].
Research Timeline
From 1869-2009, the major contributers to LCA research and consequently to retinal regeneration therapy research: "Courtesy: National Eye Institute, National Institutes of Health (NEI/NIH)."
- 1869- Dr. Theodor Leber (1840-1917), German ophthalmologist, first describes what is now known as Leber congenital amaurosis, an inherited retinal disease that causes severe visual impairment early in childhood [52]
- 1932-34- George Wald, Ph.D. first identified vitamin A in the retina during a National Research Council fellowship in biology [52]
- 1965- Human adeno-associated virus (AAV) was discovered.[52]
- 1984- Drs. Nicolas Muzyczka and Paul Hermonat publish an article on adeno-associated virus (AAV) expresses that it can be used to introduce foreign DNA into human and murine tissue culture cells.[52]
- 1990- Dr. T. Michael Redmond of the NEI's Laboratory of Retinal Cell and Molecular Biology, Section on Gene Regulation began work on RPE-specific monoclonal antibody.[52]
- 1993- Dr. T. Michael Redmond of the NEI's Laboratory, cloned RPE65, a protein necessary for processing vitamin A in the visual cycle.[52]
- 1997- RPE65 gene mutations identified as the cause of congenital blindness in some children with Leber congenital amaurosis.[52]
- 1998- Dr. T. Michael Redmond's team in the NEI's Laboratory use the knockout mouse model to establish RPE65’s role in vitamin A metabolism. RPE65 gene mutation was discovered as causing congenital blindness in Briard dogs.[52]
- 2001- Gene transfer RPE65 therapy used to restore vision in a Briard Dog by researchers at the University of Pennsylvania and University of Florida supported by the NEI.[52]
- 2005- Restored vision in Briard dog persists longer than four years following RPE65 gene transfer [52]
- 2007- NEI-supported RPE65 human clinical trial began. This study was designed to assess the safety of using a modified adeno-associated viral vector (rAAV2-hRPE65) to deliver the normal RPE65 gene to the retina.[52]
- 2009- One-year results from the NEI-supported RPE65 human clinical trial published in Human Gene Therapy and the New England Journal of Medicine. All three patients remained healthy and maintained previous visual gains. One patient also noticed a visual improvement that helped her perform daily tasks. Results from early LCA clinical studies represent one of the first steps toward the use of gene transfer therapy for an inherited form of blindness.[52]
New Research Development
While there is no effective treatment for LCA [53], a current field of research for LCA and in turn retinal restoration is that of using an altered version of the adeno-associated virus. In 2001 study on dogs with early and a similar impairment to that of LCA in children[54]. The recombinant adeno-associated virus serotype 2 (rAVV2) vector to carry the human RPE65 has restored vision in this model[55]. The virus was injected intra ocular, into 3 dogs with LCA and the results were assessed by ERG[54]. As a result of this experiment retinal function improved in eyes compared with the same eyes before treatment[54]. This experiment is a proof of concept study being the first to show gene therapy can restore vision[54]. In clinical trials undertaken in 2008 leads to improvement in visual function and the AVV has no immediate adverse effects on patients and it is suggested that these procedures would benefit children with LCA than adults[53].
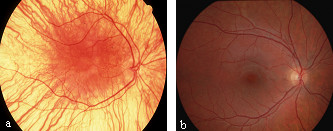
Fundus of LCA patient with RPE65 mutation
Anophthalmia and Microphthalmia
The condition of anophthalmia refers to complete absence of the globe in the presence of ocular adnexa (eyelids, conjunctiva, and lacrimal apparatus). Microphthalmia is defined as a globe with a total axial length (TAL) that is at least two standard deviations below the mean for age.[56][57][58]Anophthalmia and Micropthalmia, these two ocular manifestations have a combined incidence of approximately 2/10000 births and can be developed as unilateral or bilateral. [59] The development of both Anaophthalmia and Microphthalmia can be isolated, associated with other abnormalities or can be part of a well defined syndrome.The aetiology of these two conditions can be very complex with genetic, environmental both identified. [60][61][62]Some risks factors for these conditions are idenitifed such as maternal age over 40, multiple births, infants of low birth weight and low gestational age.[63][64][65][66]
Clinical description
Anophthalmia refers to the absence of ocular tissue in the orbit. Simple microphthalmia represent structurally normal, small eyes. The increased thickness of the sclera in these eyes are changes in blood flow are thought to be responsible for the increased incidence of uveal effusions and choroidal detachments seen. Complex micropthlamia occurs when microphthalmia is complicated with other ocular disorders. Both Anterior and posterior segment can be affected by complex microphthalmia [67]
Clinical appearance of anophthalmia and microphthalmia (upper picture: anophthalmia, lower picture:microphthalmia)
Genetic cause
There are various causes to the development of Anophthalmia and microphthalmia, the roles of several genes involved in the ocular development have been implicated. Also, some well-defined syndrome have been proved to be associated with these two conditions, such as matthew-wood syndrome and Fraser syndrom. Genes that are responsible for these conditions include, PAX-6, RAX, CHX10, SOX2, OXT2 and FOXe3. These are the main contributor to the development of anophthalmia and microphthalmia. [68][69]
SOX-2
The SOX-2 gene has been identified as the major causative gene of the two conditions, it is located on the location 3q26.3-q27. It is an important transcription factor for maintaining self-renewal, or pluripotency, of undifferentiated embryonic stem cells. [70]Although, SOX2 gene mutation is quite rare with an estimated frequency of 1 in 250,000 births, they are still considered the most common genetic cause of anophthalmia and microphthalmia to date. [71]Mutations in the SOX-2 gene is often associated with ocular malformations, they are variable in type but mostly bilateral and severe. This gene is highly expressed during lens induction process and the failure of this process may lead to anophthalmia. Also, the gene may have a role in the growth and maintenance of the developing lens. [72]SOX2 expression in humans are also seen in area of neural retina, optic stalk and lens suggesting its importance to the development of the eye.[73]
SOX2 gene also co-operates with other genes to ensure normal lens development such as the PAX 6 gene and they mutually induce each other.[74]It has been demonstrated in chicks, the co-regulation of these two gene drive lens induction in chicks, suggesting that lens induction failure in humans can be due to these genes. The lens induction is also mediated by the interaction of the two genes through their action on the γ-crystallin gene. [75]
PAX-6
The PAX-6 gene mentioned before in the Abnormal lens development section has already been stated of its importance in lens development. The mutation of this gene cause alterantion to the developing lens and the pit of the optic vesicle.[76]
RAX
The RAX gene is important for the normal eye development where it regulates the establishment and proliferation of retinal progenitor cells. Mutation of this gene will bring failure of lens induction in eye development, along with other genes such as PAX-6, CHX10, OXT2. [77][78]
FOXe3
THe FOXe3 gene is responsible for lens development in vertebrates and a mutation of this gene will lead to lens agenesis. [79][80]
Environmental
Chorioretinal Scars
Chorioretinal scars can be caused by a number of diseases during development within the uterus, it is most commonly seen after contracting the congenital varicella syndrome, rubella. syphilis, and toxoplasmosis. In rarer cases it can be seen after contraction of the herpes simplex. The Congenital Varicella Syndrome or Fetal Varicella Syndrome is the common chicken pox virus which is transferred from the mother to the fetus after she contracts the syndrome. [81] The risk of this transference is around 1.3%[82] if the mother has it during the 1st and half way through the 2nd trimester. in 1996 , seven in every 10000 pregnancies were affected by the Varicella Syndrome. [83] The reason for such a low number is because most women have usually already had the virus as a child, so their chances of getting it again are greatly reduced if not totally reduced. Fetal death caused by congenital varicella syndrome is very low at 0.8%. [84] Premature delivery is increased by 12.4% also. [85]
However, pregnancies that are affected by Congenital Varicella Syndrome are usually left with some morphological anomalies such as growth retardation(39%), gastro-intestinal lesions(23%), problems with skeletal development (68%), ocular abnomalities (68%) and many more. [86] For those born with ocular problems, the most obvious abnormality is the presence of chorioretinal scarring, but there are also many other abnormalities that are sometimes overlooked. Including atrophy of the optic discs, congenital cataracts, and Horner's Syndrome. [87]
Glossary
References
- ↑ Anthony A. Pearson The Development of the Eyelids Journal of Anatomy: 1980, 130, 1, pp. 35-42 [1]
- ↑ Ronan O'Rahilly The Prenatal Development of the Human Eye Experimental Eye Research 1975, 21, pp. 93-112 [2]
- ↑ Ronan O'Rahilly The Timing and Sequence of Events in the Development of the Human Eye and Ear During the Embryonic Period Proper Anatomy and Embryology 1983 vol:168:1 pp.87 [3]
- ↑ Ronan O'Rahilly The Prenatal Development of the Human Eye Experimental Eye Research 1975, 21, pp. 93-112 [4]
- ↑ Ronan O'Rahilly The Prenatal Development of the Human Eye Experimental Eye Research 1975, 21, pp. 93-112 [5]
- ↑ Anthony A. Pearson The Development of the Eyelids Journal of Anatomy: 1980, 130, 1, pp. 35-42 [6]
- ↑ Ronan O'Rahilly The Timing and Sequence of Events in the Development of the HUman Eye and Ear During the Embryonic Period Proper Anatomy and Embryology 1983 vol:168:1 pp.87[7]
- ↑ Ronan O'Rahilly The Prenatal Development of the Human Eye Experimental Eye Research 1975, 21, pp. 93-112 [8]
- ↑ Anthony A. Pearson The Development of the Eyelids Journal of Anatomy: 1980, 130, 1, pp. 35-42 [9]
- ↑ McAvoy, J W Lens Development Eye (The Royal College of Ophthalmologists) 1999 vol:13 3b pp.25 [10]
- ↑ Grindley, J C Et Al The role of Pax-6 in eye and nasal development 1995 vol:121:5 pp.1433 [11]
- ↑ McAvoy, J W Lens Development Eye (The Royal College of Ophthalmologists) 1999 vol:13 3b pp.25 [12]
- ↑ Grindley, J C Et Al The role of Pax-6 in eye and nasal development 1995 vol:121:5 pp.1433 [13]
- ↑ McAvoy, J W Lens Development Eye (The Royal College of Ophthalmologists) 1999 vol:13 3b pp.25 [14]
- ↑ Mihelec, M, St Heaps, L, Flaherty, M Chromosomal rearrangements and novel genes in disorders of eye development, cataract and glaucoma.Twin Research and Human Genetics 2008 vol:11 pp.412-21 [15]
- ↑ Glaser, T Et al Genomic structure, evolutionary conservation and aniridia mutations in the human PAX6 gene Nature Genetics 1992 vol:2:3 pp.232 [16]
- ↑ Grindley, J C Et Al The role of Pax-6 in eye and nasal development 1995 vol:121:5 pp.1433 [17]
- ↑ Grindley, J C Et Al The role of Pax-6 in eye and nasal development 1995 vol:121:5 pp.1433 [18]
- ↑ Hejtmancik, JF, Congenital Cataracts and their Molecular Genetics 2008 Semin Cell Dev Biol, vol:19:2,pp.134–149.[19]
- ↑ Zilinski C, Brownell I, Hashimoto R et al Expression of FoxE4 and Rx visualizes the timing and dynamics of critical processes taking place during initial stages of vertebrate eye development. Dev Neurosci 2004 vol:26:5-6), pp. 294-307.[20]
- ↑ Zilinski C, Brownell I, Hashimoto R et al Expression of FoxE4 and Rx visualizes the timing and dynamics of critical processes taking place during initial stages of vertebrate eye development. Dev Neurosci 2004 vol:26:5-6), pp. 294-307.[21]
- ↑ Hejtmancik, JF, Congenital Cataracts and their Molecular Genetics 2008 Semin Cell Dev Biol, vol:19:2,pp.134–149.[22]
- ↑ Cohen, D, Bar-Yosef, U, Levy, J, Gradstein, L, Belfair,N et al, Homozygous CRYBB1 Deletion Mutation Underlies Autosomal Recessive Congenital Cataract 2007, Invest. Ophthalmol. Vis. Sci, vol:48:5 pp. 2208-2213[23]
- ↑ Cohen, D, Bar-Yosef, U, Levy, J, Gradstein, L, Belfair,N et al, Homozygous CRYBB1 Deletion Mutation Underlies Autosomal Recessive Congenital Cataract 2007, Invest. Ophthalmol. Vis. Sci, vol:48:5 pp. 2208-2213[24]
- ↑ Hejtmancik, JF, Congenital Cataracts and their Molecular Genetics 2008 Semin Cell Dev Biol, vol:19:2,pp.134–149.[25]
- ↑ Hejtmancik, JF, Congenital Cataracts and their Molecular Genetics 2008 Semin Cell Dev Biol, vol:19:2,pp.134–149.[26]
- ↑ Ronan O'Rahilly The Timing and Sequence of Events in the Development of the HUman Eye and Ear During the Embryonic Period Proper Anatomy and Embryology 1983 vol:168:1 pp.87 [27]
- ↑ Ronan O'Rahilly The Timing and Sequence of Events in the Development of the HUman Eye and Ear During the Embryonic Period Proper Anatomy and Embryology 1983 vol:168:1 pp.87 [28]
- ↑ Jiao X, Sultana A, Garg P, Ramamurthy B, Vemuganti GK et al Autosomal recessive corneal endothelial dystrophy (CHED2) is associated with mutations in SLC4A11.J Med Genet. 2007, vol:44:1, pp64-8[29]
- ↑ Hemadevi B, Veitia RA, Srinivasan M, Arunkumar J et al Identification of mutations in the SLC4A11 gene in patients with recessive congenital hereditary endothelial dystrophy. Arch Ophthalmol. 2008 vol:126:5 pp.700-8.[30]
- ↑ Schmedta,T,Silvac,MM, Ziaeia, A, Jurkunas, U, Molecular bases of corneal endothelial dystrophies 2012, vol:95,pp. 24-34[31]
- ↑ Jiao X, Sultana A, Garg P, Ramamurthy B, Vemuganti GK et al Autosomal recessive corneal endothelial dystrophy (CHED2) is associated with mutations in SLC4A11.J Med Genet. 2007, vol:44:1, pp64-68[32]
- ↑ Gröger N, Fröhlich H, Maier H, Olbrich A, Kostin S, Braun T, Boettger T. SLC4A11 prevents osmotic imbalance leading to corneal endothelial dystrophy, deafness, and polyuria. J Biol Chem 2010 vol:7:19, pp.14467-74.[33]
- ↑ Vithana,EN, Morgan, P, Sundaresan, P, Ebenezer, ND et al Mutations in sodium-borate cotransporter SLC4A11 cause recessive congenital hereditary endothelial dystrophy (CHED2) 2006 Nature genetics, vol:38, pp.755-757[34]
- ↑ Vithana,EN, Morgan, P, Sundaresan, P, Ebenezer, ND et al Mutations in sodium-borate cotransporter SLC4A11 cause recessive congenital hereditary endothelial dystrophy (CHED2) 2006 Nature genetics, vol:38, pp.755-757[35]
- ↑ <pubmed>15231395</pubmed>
- ↑ <pubmed>15231395</pubmed>
- ↑ <pubmed>14625556</pubmed>
- ↑ <pubmed>14625556</pubmed>
- ↑ <pubmed>14625556</pubmed>
- ↑ <pubmed>PMC2940541</pubmed>
- ↑ <pubmed>9775209</pubmed>
- ↑ <pubmed>9775209</pubmed>
- ↑ <pubmed>9775209</pubmed>
- ↑ <pubmed>9775209</pubmed>
- ↑ <pubmed>9775209</pubmed>
- ↑ <pubmed>9775209</pubmed>
- ↑ 48.0 48.1 http://www.blindness.org/index.php?view=article&catid=38%3Aother-retinal-diseases&id=253%3Aleber-congenital-amaurosis&option=com_content&Itemid=88
- ↑ http://www.springerlink.com/content/rmj766pjrq130011/
- ↑ 50.0 50.1 50.2 50.3 50.4 50.5 <pubmed>15231395</pubmed>
- ↑ 51.0 51.1 51.2 51.3 51.4 <pubmed>PMC3283211</pubmed>
- ↑ 52.00 52.01 52.02 52.03 52.04 52.05 52.06 52.07 52.08 52.09 52.10 52.11 http://www.nei.nih.gov/lca/timeline.asp
- ↑ 53.0 53.1 <pubmed>18441371</pubmed>
- ↑ 54.0 54.1 54.2 54.3 <pubmed>11326284</pubmed>
- ↑ <pubmed>PMC2940541</pubmed>
- ↑ Bardakjian,TM, Weiss,A, Schneider, AS Anophthalmia / Microphthalmia Overview 2004 GeneReviews [36]
- ↑ Bardakjian,TM, Schneider, A, The genetics of anophthalmia and microphthalmia, 2011 Current Opinion in Ophthalmology, vol:22, pp.309 – 313[37]
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [38]
- ↑ Bardakjian,TM, Schneider, A, The genetics of anophthalmia and microphthalmia, 2011 Current Opinion in Ophthalmology, vol:22, pp.309 – Microphthalmia is reported in 3.2 – 11.2% of blind children [7].313[39]
- ↑ Bardakjian,TM, Weiss,A, Schneider, AS Anophthalmia / Microphthalmia Overview 2004 GeneReviews [40]
- ↑ Bardakjian,TM, Schneider, A, The genetics of anophthalmia and microphthalmia, 2011 Current Opinion in Ophthalmology, vol:22, pp.309 – 313[41]
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [42]
- ↑ Shaw GM, Carmichael SL, Yang W, Harris JA, Finnell RH, Lammer EJ, Epidemiologic characteristics of anophthalmia and bilateral microphthalmia among 2.5 million births in California,1989–1997. Am J Med Genet A 2005, vol:137, pp.36-40
- ↑ Kallen B, Robert E, Harris J: The descriptive epidemiology of anophthalmia and microphthalmia. Int J Epidemiol 1996, vol:25, pp.1009-1016.
- ↑ Forrester MB, Merz RD: Descriptive epidemiology of anophthalmia and microphthalmia, Hawaii, 2006 Birth Defects Res A Clin Mol Teratol vol:76, pp.187-92.
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [43]
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [44]
- ↑ Bardakjian,TM, Schneider, A, The genetics of anophthalmia and microphthalmia, 2011 Current Opinion in Ophthalmology, vol:22, pp.309 – 313[45]
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [46]
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [47]
- ↑ Slavotinek, AM, Eye development genes and known syndromes 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[48]
- ↑ Ragge NK, Lorenz B, Schneider A, Bushby K, de Sanctis L et al SOX2 anophthalmia syndrome. 2005 Am J Med Genet A vol:135(1), pp.1-7[49]
- ↑ Slavotinek, AM, Eye development genes and known syndromes 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[50]
- ↑ Slavotinek, AM, Eye development genes and known syndromes 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[51]
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [52]
- ↑ Slavotinek, AM, Eye development genes and known syndromes 2011,Molecular Genetics and Metabolism, vol:104(4), pp.448-456[53]
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [54]
- ↑ Bardakjian,TM, Schneider, A, The genetics of anophthalmia and microphthalmia, 2011 Current Opinion in Ophthalmology, vol:22, pp.309 – 313[55]
- ↑ Verma,AS, FitzPatrick, DR,Anophthalmia and microphthalmia 2007 Orphanet Journal of Rare Diseases,pp.2-47 [56]
- ↑ Bardakjian,TM, Schneider, A, The genetics of anophthalmia and microphthalmia, 2011 Current Opinion in Ophthalmology, vol:22, pp.309 – 313[57]
- ↑ Lambert, S et al Ocular Manifestations of the Congenital Varicella Syndrome Archives of Ophthalmology 1989 vol:107:1 pp.52 [58]
- ↑ Boussault, P et al Chronic Varicella-Zoster Skin Infection Complicating the Congenital Varicella Syndrome Pediatric Dermatology 2007 vol:24:4 pp. 429 [59]
- ↑ Dufour, P et al. Varicella and Pregnancy European Journal of Obstetrics & Gynecology and Reproductive Biology, 1996 vol:66:2 pp.119 [60]
- ↑ Dufour, P et al. Varicella and Pregnancy European Journal of Obstetrics & Gynecology and Reproductive Biology, 1996 vol:66:2 pp.119 [61]
- ↑ Dufour, P et al. Varicella and Pregnancy European Journal of Obstetrics & Gynecology and Reproductive Biology, 1996 vol:66:2 pp.119 [62]
- ↑ Dufour, P et al. Varicella and Pregnancy European Journal of Obstetrics & Gynecology and Reproductive Biology, 1996 vol:66:2 pp.119 [63]
- ↑ Lambert, S et al Ocular Manifestations of the Congenital Varicella Syndrome Archives of Ophthalmology 1989 vol:107:1 pp.52 [64]
- ↑ Dufour, P et al. Varicella and Pregnancy European Journal of Obstetrics & Gynecology and Reproductive Biology, 1996 vol:66:2 pp.119 [65]
- ↑ Boussault, P et al Chronic Varicella-Zoster Skin Infection Complicating the Congenital Varicella Syndrome Pediatric Dermatology 2007 vol:24:4 pp. 429 [66]
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
--Mark Hill 12:22, 15 August 2012 (EST) Please leave the content listed below the line at the bottom of your project page.
2012 Projects: Vision | Somatosensory | Taste | Olfaction | Abnormal Vision | Hearing