2012 Group Project 4
Olfaction Development
Introduction
WAKE UP AND SMELL THE ROSES! The sense of smell, or otherwise known as Olfaction is the sense mediated by sensory cells located in the nasal cavity. Chemoreceptors within the naval cavity are activated by chemicals in the air which are known as odorants. Odorants produce olfactory sensation at very low concentration, and through the reaction with chemoreceptors enables the sense of smell in humans. The olfactory system are often divide into a peripheral mechanism, activated by an external stimulus and transforming it into an electric signal in neurons, and a central mechanism where all signals formed by olfactory are integrated in the central nervous system and processed to recognise odor. Over 1000 genes which make up three percent of the total human genome which encode for olfactory receptor types which can each detect a small number of related molecules and respond with different level of intensity. It has been discovered that olfactory receptor cells are highly specialized to particular odors.
This page seeks to explore the development of the olfactory system in addition to their function and physiology. This page will also examine both structural and neurological abnormalities that can arise. Within this page, current research is looked into, and analyzes the possible future research within the olfactory system.
History of Discovery
| Year | Person | Contribution |
| 1703 | Frederick Ruysch |
Discovery of Vomeronasal organ [1] |
| 1856 | Maestre de San Juan | The first person to link hypogonadism to the olfactory system[2] |
| 1891 | Von Kupffer | Von Kupffer is recognised with description of the olfactory placodes as ectodermal thickenings[3]. For a time they were termed Kupffer placodes. |
| 1899 | B.H. Buxton | B. H. Buxton published a paper containing a series of photographs of a day 25 human embryo in the Journal of Anatomy and Physiology. He noted the thickened ridges of epiblast, the olfactory plates but there were at that stage no olfactory pits [4]. |
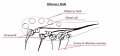
| 1900s | Julius Kollmann | 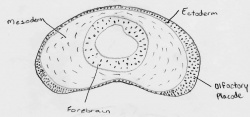 Kollmann's diagram of developing nasal placode[1]
|
| 1941 | Anthony A. Pearson | Pearson conducted a study in examining serial sections of human embryos to understand the development of the olfactory nerve. It was seen the cells migrate from the olfactory epithelium up obliquely toward the brain collecting as fibers. His research indicated that olfactory nerve fibers start to form communications with the brain six weeks into development. He also asserted that the olfactory bulb starts to form in a 17mm embryo following which the proximal end of the olfactory nerve forms a sheath of fibers over the bulb. The fibers of this sheath collect together and continue to develop to form the fila olfactoria which eventually pass through the cribriform plate[7] |
| 1944 | Frank Kallmann | Kallmann looked at three families who suffered from the now-called Kallmann's Syndrome. Frank Kallmann was a geneticist and psychiatrist. By analysing these familial groups he hypothesised about the inheritance of the disease[2] |
| 1954 | De Morsier | De Morsier reported other patients suffering from similar symptoms to those reported by Kallmann (hypogonadism, anosmia and midline anatomic defects) but he termed the condition olfactogenital dysplasia suggesting a link between hypogonadism and the hypothalamus[2] |
| 2004 | Linda B. Buck
and Richard Axel |
Won the Nobel Prize in Physiology or Medicine for revealing the large number of genes involved in odour reception. [8] |
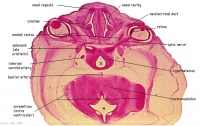
Anatomy of the Olfactory System
Nasal Cavity
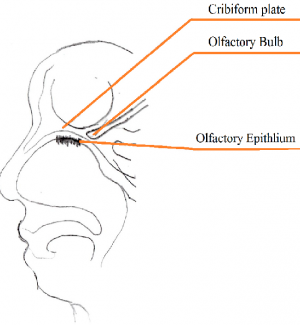
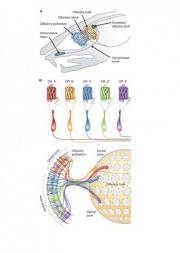
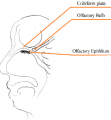
The nasal cavity is an important structure of the Olfactory system as within the turbinates or nasal conchae are found. These structures act to direct air inspired toward the olfactory epithelium. The epithelium is located in the upper posterior region of the nasal cavity and is approximately a couple of centimeters wide. Olfactory epithelium is a specialized epithelium which contains around 100 million receptor cells. The olfactory epithelial cells is also the origin of olfactory vesicles which are known to contain kinocilia. The Olfactory vesicles are also known to serve in the process of stimulus transduction.
Olfactory Epithelium
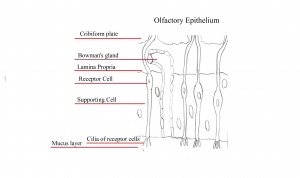
Olfactory epithelium consists of pseudostratified epithelium which contain olfactory receptors along with nerve cells whose axons attach to the olfactory bulb of the brain. It consists of 3 different cell types, namely basal, supporting and olfactory receptor cells as shown in the diagram to the right. Basal cells are stem cells which give rise to olfactory receptor cells. [9]. The continuous supply of neurons and the replacement of neurons by less differentiated stem cells is unique only to the olfactory system. The third type of cells is the supporting cells which are found among the receptor cells and their function is to empty their content onto the mucosal surface using their microvili and secretory granules.[10].
Olfactory Bulb
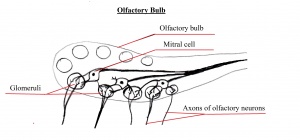
The olfactory bulb is a structure located in the forebrain which receives neural information about odours detected by recepotor cells within the nasal cavity. The information is extended to the olfactory bulb by the axons of olfactory receptor where the information is processed, and the smell of the odour determined. The olfactory bulb is essential for olfaction as it transmits information from the olfactory epithelium and up to the brain. The bulb receives input from olfactory nerves which constitutes the axons of olfactory receptor neurons. [11].
Structures known as glomeruli form by a number of olfactory axons joining together such that each glomerulus obtain information from olfactory neurons which have the identical odour receptors. These glomeruli structures are also surrounded by dendrites belonging to mitral cells which transmit electrical signals to the olfactory cortex in the brain.[12].
Cribiform plate
The cribiform plate is composed of the ethmoid bone and acts as the roof of the nasal cavity. The plate is narrow and deeply grooved as it acts as a support structure for the olfactory bulb, and pierced by a foramina to allow the passage of olfactory nerves through the plate. The foramina located in the middle of the grove allows the passage of nerves through the roof of the nasal cavity, whereas the the foramina on the medial parts of the groove allows the passage of nerves to the superior part of nasal septum. The foramina located on the lateral side of the grove also permits the passage of nerves and direct them towards superior nasal concha. If the cribriform plate happens to get fractured, it can lead to the loss of sense of smell and the leaking of cerebrospinal fluid into the nose. [13].
Normal Function
Olfactory Signal Transduction
Olfactory Signal Transduction is initiated by any substance that emit molecules known as odours. The olfactory transduction is dependent upon the dissolving of these odorants in the mucus layer of the olfactory epithelium in order to bind to specific chemoreceptors in order for chemical signals to be converted into electrical signals. The transformation into electrical signal is essential for signal transduction for the brain to perceive the initial odourants as smell. [14].
When odorant molecules bind to receptors in olfactory epithelium, a G protein coupled receptors known as G(αolf) is activated which then happen to activate adenylate cyclase, an enzyme which catalyses the formation of cyclic AMP (cAMP). In most receptor cells, cAMP acts as a second messenger, however in the olfactory system cAMP bind to cation channels which permits sodium and calcium ions by binding to and opening cyclic nucleotide gated ion channel to travel through the membrane and enter the cell. [15]. The main effect of ion entry into the cell is depolarisation, and activation of chloride channels resulting in greater depolarisation by the efflux of chloride ions. If the depolarization in the cell is great enough, an action potential is generated on the axon of the receptor cell and transferred to the brain through the olfactory bulb. [16]
Similar to other G-protein mediated pathways, the olfactory sensory neuron is exposed to negative feedback by the cAMP cascade activated by odours. The negative feedback loop has been discovered to be responsible for the adaption of odours and deactivation of response after exposure for a certain period of time. [17].
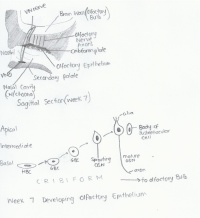
Timeline of developmental process
| Week/Stage | Patterning Genes | Description | Image |
| Week 4 |
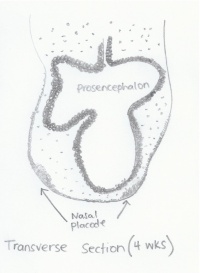
Early expression of genes in the Hes5 family suggests it's role in pre-patterning of the placode ectoderm.[18] Wnt, BMP, and FGF are known to play a role in the early migration of neural crest cells to the olfactory placode however the exact mechanisms of signalling are still uncertain.[19] |
An interesting study of neural crest cell migration in rats revealed that the origins of neural crest cells during frononasal development change in relation to the stage of somite development[21]: - 3 to 5 somite stage: lateral edge of the prosencephalon produced cells which migrated to the frontonasal mass while anterior neural crest cells in the prosencephalon contributed to the nasal placode epithelium. Mesencephalic region produced neural crest cells which contributed to the frontonasal mass.[21] - 5 to 10 somite stage: Anterior portion of the mesencephalon continued producing crest cells for migration to the frontonasal mass.[21]
|
|
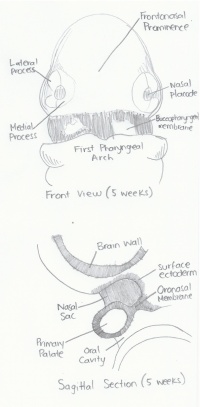
| Week 5 |
Early Notch signalling plays an important role in the inhibiton of early differentiation of olfactory epithelial cells into olfactory sensory neurons.[25] |
|
|
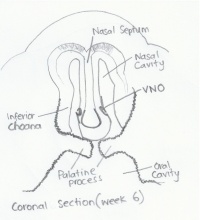
| Week 6 |
FGF plays a role in signalling the generation of olfactory sensory epithelium. [27] |
The superior, middle, and inferior nasal conchae develop as the lateral walls of the nasal cavities proliferate and so elevate.[19]
|
|
| Week 7 |
BMP plays a role in signalling the generation of non-sensory epithelium.[27] Neurog1 and Neurog2 play a role in signalling the specialisation of the olfactory epithelium and olfactory bulb neuron morphogenesis.[31] |
After birth, it functions to drain excess tears from the conjunctiva of the eye into the nasal cavity. [19]
The lining of the olfactory placode differentiates into three layers within the pseudo-stratified organised epithelium:[28] - Inner basal layer: composed of two cell types, the horizontal and globose basal cells.[28] - Intermediate layer: contains olfactory sensory neurons which ascend to the apical layer as they become more differentiated.[28] - Apical layer: contains the mature olfactory sensory neurons as well as the nuclei and bodies of the supporting sustentacular cells. [28]
The synpasing cells differentiate to become the secondary sensory neurons, mitral cells, of the olfactory pathways. [30]
As a result, the CNS olfactory tracts look stalk-like.
|
|
| Week 8/Fetal development |
BMP plays a role in signalling the generation of non-sensory epithelium.[27] |
The degeneration of the VNO is noted by the closure of the duct and formation of a cyst like structure in the nasal septum. However, in most adults, the duct is left opened to a certain degree.[30]
|
Also see normally developed sensory structures of olfaction
|
Congenital Abnormalities
Olfactory Defects
Anosmia is defined as the absence of a sense of smell. Hyposmia refers to a reduced sense of smell. These conditions, when they occur as a congenital feature, can be associated with Choanal Atresia or Kallmann Syndrome. At present, these conditions are the most commonly recognised congenital conditions contributing to abnormal olfactory function.
Choanal Atresia
Introduction and Epidemiology
Choanal atresia is a congenital abnormality characterised by narrowing or complete obliteration of the nasal aperture by a bony or membranous occlusion.[34][35] This anomaly occurs in 1 in every 7000 to 8000 births with a female predominance.[36]
- 45% of cases are bilateral involving both choanae [35]
- Mixed bony and membranous anomalies were most common (70%) followed by pure bony atresia (30%) with no pure membranous anomalies[35].
Pathophysiology
At present, the exact cause of choanal atresia is still under debate. The table below displays models which have been proposed to explain how choanal atresia may occur in the developing human.
| Risk Factor Model | Description |
| Embryonic | Ramsden [37]notes that "A number of embryological models for the development of choanal atresia have been proposed, although none of them are wholly supported by convincing clinical evidence": |
| Genetic |
|
| Molecular |
|
Clinical Features
Neonates are obligate nose breathers, hence neonatal nasal obstruction as seen in choanal atresia is a serious deformity.[48] In order to avoid severe hypoxia and death, immediate diagnosis and intervention are key.[48] The severity of the clinical features of choanal atresia depends on the whether the obstruction is unilateral or bilateral.[37]
Bilateral
- At birth present with asphyxia neonatorum: pathological changes caused by hypoxia from affected respiration. This is a medical emergency requiring an oral airway tube or intubation then immediate surgical intervention [37][49]
- Obvious airway obstruction [37]
- Stridor, a harsh vibrating sound when breathing [37]
- Paradoxical cyanosis: cyanosis (blue appearance from deoxygenated blood) is present in the infant at rest but improves with exertion such as crying [37]
Unilateral
- Not as life threatening as bilateral choanal atresia; more often diagnosed in childhood than in infancy [37][49]
- Mucoid rhinorrhea, constant mucous fluid discharge from nose [37][49]
- Dysosmia: distorted olfaction [49]
- Obstructive sleep apnoea [49]
Kallmann Syndrome
Introduction and Epidemiology
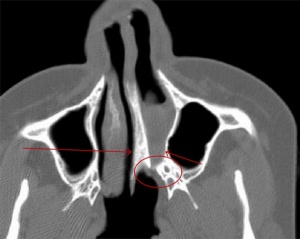
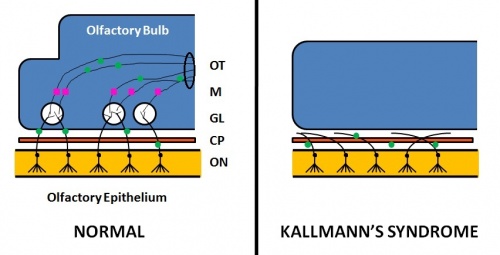
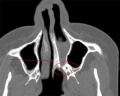
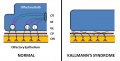
Kallmann syndrome is a clinically and genetically heterogeneous disorder, described as a hypogonadotropic hypogonadism characterized by a diminished or absent sense of smell.[50] [51] The incidence of Kallmann syndrome is uncertain but is estimated to occur in 1 in 10,000 to 1 in 50,000 people[2], affecting males to females in a 5:1 ratio. [52] Anosmia or hyposmia occurs as a results of impaired development of the olfactory bulbs and olfactory nerves. [52] Additionally, hypogonadism results due to the reduced production of Gonadotropin-releasing hormone (GnRH). Kallmann syndrome can be inherited as an X-linked recessive, autosomal dominant or autosomal recessive trait.[52]
Pathophysiology
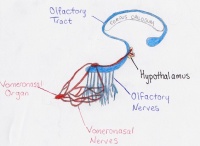
The olfactory bulb (OB) is the first neuronal checkpoint for olfactory information.[53] The OB receives and processes sensory inputs from olfactory receptor neurons embedded in the olfactory epithelium and then transmits the information to the olfactory cortex.[53] During embryonic development, axons from the olfactory receptor neurons exit the olfactory epithelium, grow toward the brain, and penetrate the OB where they synapse with the dendrites of mitral cells. [53] The axons of these neurons form the olfactory tract. In Kallmann syndrome, the OB is abnormal in structure or not present; coupled with neuronal migration failures, olfactory signals from the environment are prevented from being transmitted to the cerebral cortex. Additionally, the failure of the GnRH neuronal migration to the hypothalamus results in a loss of a key path in the negative feedback loop for sex hormone production.
Genetic Factors In Kallmann syndrome, there are distinct abnormalities in the OB development arising due to the abnormal or lack of expression of certain proteins and genes. Kallmann syndrome can be X-linked , autosomal dominant or autosomal recessive.[52] To date, mutations in six genes and the proteins they encode (see table below) have been attributed to Kallmann syndrome, though their functions are still being researched. However, only 30% of patients with a clinical diagnosis of Kallmann Syndrome are found to have a mutation in these genes.[54] Note that CHD7 can also be mutated in Kallmann's syndrome patients, though these individuals tend to have additional features that are part of the CHARGE syndrome phenotype described in the section on Choanal Atresia.[55]
| Gene | Mode of Inheritance | Role in Kallman’s Syndrome |
| KAL1 | X-linked recessive | KAL1 normally encodes glycoprotein anosmin 1 and is expressed in the outer neuronal layers of the developing olfactory bulb.[56] Anosmin-1 stimulates lateral olfactory tract axon branching and outgrowth from the OB towards the piriform cortex; this is through patterning of the mitral and tufted cell axons to the olfactory cortex.[53] Consequently, in the absence of anosmin-1, Kallmann syndrome arises due to abnormal olfactory neuronal development.[53] Additionally, anosmin-1 has been shown to interact with FGFR1, explaining the digenic nature of Kallmann syndrome.[57] |
| FGFR1 (KAL2) | Autosomal-dominant [57] | FGFR1 encodes fibroblast growth factor receptor 1 involved in OB morphogenesis and GnRH neuronal development and migration.[57] [58] When absent, Kallmann syndrome arises due to agenesis or digenesis of the olfactory bulb and failure of GnRH neuronal development and migration. |
| FGF8 | Autosomal-dominant [57] | Encodes the key ligand for FGFR1. FGF binds with high affinity to FGFR and induces receptor activation. The absence of FGF8 produces a similar phenotype to the KAL2 mutation as the receptor is not activated.[57] |
| PROKR2 (KAL3) | Monoallelic PROKR2 mutations are not sufficient to produce the disease phenotype; it is hypothesised that digenic or oligogenic inheritance of KS in patients heterozygous for PROKR2 mutations produce the disease phenotype[59][60]. | Encodes the G protein-coupled receptor prokineticin receptor-2 which is known to be involved in intracellular Ca2+ signalling.[60] However, the exact role in Kallmann syndrome has yet to be clarified.[60] |
| PROK2 (KAL4) | Hypothesised to have mendelian autosomal recessive transmission in addition to oligogenic transmission.[59] | Encodes the PROKR2 ligand.[60] When PROK2 mutated, the ligand is not expressed preventing prokineticin receptor-2 activation; this produces similar effects to PROKR2 abnormalities. |
Clinical Features
Kallmann Syndrome has the classical hypogonadotropic hypogonadism (HH) feature of an absence of puberty but is distinguished from other HH syndromes by an affected sense of smell[50]. There exists additional characteristics that are not specific to Kallmann syndrome but may aid in correct diagnosis of this particular HH[50]. The following characteristics of Kallmann syndrome may or may not be present in different cases, often varying according to genotype [50]:
Reproductive Features
- Hypogonadotropism leading to failed or arrested puberty [61]
- Hypogonadism
- Cryptorchidism: Failure of one or both testes to migrate into the scrotum during male foetus development[61]
- Gynaecomastia: The development of abnormal mammary glands in males characterised by enlarged breasts[61]
- Amennorhoea: the absence of menstruation, in females[61]
Non-Reproductive Features
- Affected sense of smell: decreased (hyponosmia) or absent (anosmia) sense of smell. [61] Anatomically, the olfactory bulbs and olfactory tracts demonstrate aplasia or hypoplasia. [61]
- Eunuchoidism bone structure, defined by long limbs as a result of inadequate calcification
- Unilateral renal aplasia [62]
- Cleft palate[61]
- Pes cavus: Also called clawfoot, refers to a deformity of the foot characterised by an overexaggerated arch and hyperextension of the toes. [61]
- Neurological symptoms
- Synkinesia: Patients can conduct voluntary movements, however, with accompanied involuntary muscular movements.
- Abnormalities in eye movement
- Cerebellar ataxia: Reduced control over muscle coordination arising from defects or damage to the cerebellum.
- Evoked horizontal nystagmus: fast involuntary movements of the eyes that may impair vision. Can be described as a "rapid flicking side to side" movement.
- Sensorineural deafness
- Spatial attentional abnormalities
- Spastic paraplegia characterised by stiffness and contraction in the lower limbs as a result of neuronal dysfunction.
- Mental retardation [61][63] [64] [65]
Diagnosis and Treatment
| Diagnosis and Treatment |
|---|
| Diagnosis
Due to the low incidence of Kallmann syndrome, correct diagnosis is often delayed, despite early childhood signs such as anosmia and cryptorchidism. [66] Instead, doctors often dismiss Kallmann syndrome as constitionally delayed puberty. [66] Other differential diagnoses include potential presence of hypothalamic or pituitary tumours.[66] Due to the varied phenotype and genotype of Kallmann, multiple tests are required in order to properly diagnose the syndrome. The following diagnostic tests are often employed:
|
| Treatment |
Current Research
Contribution of Neural Crest and Ectoderm to Nasal Placode
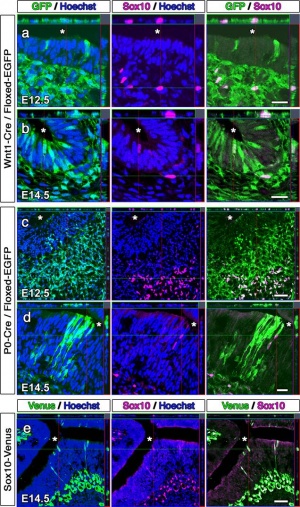
Forni PE et al.published last year explored the individual neural crest and ectodermal contributions to the nasal placode through the use of genetic Cre-lox tracing in two mice species. One mouse species was Wnt1Cre, a neural-crest specific line. The other species was Crect, an ectodermal specific line. The Cre-lox genetic tracing of the two species determined that olfactory ensheathing cells are neural crest in origin. Neural crest was also shown to contribute to cells of the olfactory epithelium and vomeronasal organ along with GnRH-1 neurons. The findings of this paper allowed provided an understanding of the link relating neural crest defects to diseases such as anosmia and Kallmann syndrome[67].
Contribution of Cranial Neural Crest to Olfactory System
The dual origin of the peripheral olfactory system also investigated the contribution of cranial neural crest cells in olfaction development used transgenic mice. The neural crest cells of these mice permanently express green fluorescent protein (GFP) which allowed them and their descendants to be traced. Analysis showed GFP-positive cells in the olfactory epithelium, olfactory ensheathing cells . Similar analysis of chick embryos demonstrated dissociated cells of the olfactory mucosa which displayed the ability to self-renew, suggesting the presence of neural crest progenitors in the olfactory mucosa. The paper concluded that the cranial neural crest contributed a larger portion than previously thought to the olfaction system and may be accountable for the olfactory epithelium’s ability to regenerate[68].
Specialisation of Olfactory Bulb and Epithelium Reliant on Specific Genes
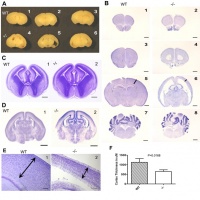
Shaker T. et al. published a paper in August this year looking into the effect of genes Neurog1 and Neurog2 on cell specialisation in the olfactory bulb and olfactory epithelium. It was concluded that Neurog1 and Neurog2 are both necessary for the development of the olfactory system and are reliant on interactions between the olfactory bulb and olfactory epithelium. One particular section of the research looked to determine whether Neurog1 and Neurog2 were required for olfactory bulb development. A loss-of-function technique was utilised to compare single and double null mutants. It was concluded that Neurog1 is required for correct growth and lamination of the olfactory bulb and that Neurog1 and Neurog2 are required for overall bulb morphogenesis[69].
Migratory Path of GnRH
Another paperalso published this year examined the migratory path of Gonadtropin-releasing hormone (GnRH) neurons and how this path is modulated by members of the Slit-Robo group of ligand ligand-receptors. Gonadtropin-releasing hormone neurons originate in the nasal placode and migrate by the olfactory and vomeronasal axons to the hypothalamus in the forebrain. GRH is responsible for regulation of reproduction in mammals. Deficiency in it causes hyopgonadotropic hypogonadism and Kallmann syndrome. The current study used genetically altered mouse models to demonstrate the role of Slit2 and Robo3 in GnRH migration. Mice lacking Slit2 were found to have fewer GnRH neurons compared to wild type mice with Slit2[70]
SEMA3A deletion and Kallmann syndrome
A recent study[71] published in the Oxford Medicine's Human Reproduction Journal sought to identify new genes responsible for Kallmann's syndrome(KS) by conducting a comparative genomic hybridization array on KS patients with no mutations in known KS genes. A family with a history of KS was involved in the study and lead to the discovery of a heterozygous deletion at locus 7q21.11. Further investigation found that this was a deletion of the gene SEMA3A. SEM3A codes for semaphorin 3A, a protein that interacts with neuropilins: transmembrane glycoprotein receptors in neurons[71]. Moreover, analysis of the pattern of KS incidence in the family in conjunction with genetic testing found the mutation to be autosomal dominant[71]. In order to consolidate the link between SEMA3A deletion and KS, the study looked to the literature. It was found that studies with semaphorin 3A-knockout mice have a KS phenotype: abnormal migration of GnRH neurons to the hypothalamus as a result of faulty signal transduction[71].
Colony Stimulation Factor-1 Receptor and Embryonic Olfactory Development
A study by Erblich et al. [72] sought to study the transmembrane tyrosine kinase receptor for colony stimulating factor-1 (CSF-1R). Mice homozygous for a null mutation (-/-) in the Csflr gene as well as mice homozygous for non-mutated Csflr (+/+) were utilised to study CSF-1R function. Antibody staining for CSF-1R in +/+ mice showed expression of CSF-1R in the microglia but not in the astrocytes, neurons or glial cells. In contrast, the -/- mice showed no CSF-1R expression at all. Moreover, cell counts showed that in -/- mice, the numbers of microgliadeclined within three weeks of birth. The microglia depletion in -/- mice was accompanied by abnormal structural integrity of the brain: there was significant ventricular enlargement with reduced parenchymal volume. From these findings, it is apparent that CSF-1R has an important role in microglial development and normal brain architecture. In regards to the olfactory bulb, there was an apparent reduction in size for the -/- mice but no obvious change in external structure. However, the olfactory bulb was hollowed out in the -/- mice as a result of the enlarged cerebrospinal fluid compartment impinging on the olfactory bulb's normal growth. Testing for olfactory deficits revealed that an absence of Csf1r gene is anosmic. These findings show that CSF-1R is required for the function and integrity of the olfactory system.[72]
Lhx2-dependent Integration of Olfactory, Vomeronasal, and GnRH Neurons
The LIM-homeodomain 2 gene (Lhx2) is normally expressed in the forebrain, the olfactory bulb, the olfactory sensory neurons (OSNs) and the vomeronasal sensory neurons in the developing human (VSNs)[73]. When Lhx2 is not expressed, specification of olfactory sensory neurons (OSNs) becomes abnormal[73]. A study[73] published in 2012 sought to identify the exact consequences of absent Lhx2-dependent OSN specification on the development of the primary olfactory pathway. The method involved utilising transgenic mice with inactivated Lhx2 gene in OSNs but not in VSNs the olfactory bulb. The study found that Lhx2-dependent OSN specification is essential for synapses between OSN and target neurons in the olfactory bulb. Moreover, the mutant phenotype showed that expansion of the olfactory bulb is dependent on innervation by OSNs expressing Lhx2. Additionally, Lhx2-dependent maturation of OSNs is required for formation of the vomeronasal nerve and the migration of gonadotropin-releasing hormone (GnRH) cells toward the developing hypothalamus. The implications of these findings to olfactory research are a further understanding of the innervation mechanisms of the olfactory bulb during development. Moreover, the findings of the study can aid in understanding congenital olfactory defects such as those seen in Kallmann syndrome.
Glossary
Aplasia: Absent development of an organ or tissue.
Anosmia: Lack of smell.
Cerebellar ataxia: Reduced control over muscle coordination arising from defects or damage to the cerebellum.
Cribiform Plate: The inferior surface of the ethmoid bone which roofs the nasal cavities.
Ectoderm: The outermost layer of the trilaminar embryo. Differentiates to form structures including the epidermis and neural tissue.
Epiblast: The collective term for embryonic mesoderm and ectoderm before differentiation
Eunuchoidism: Male hypogonadism characterised by the failure of the testes to develop and an absence of secondary sexual characteristics.
Frontonasal Prominence: An ectodermal expansive process that arises during the third week of embryonic development and forms the forehead and nasal bridge.
Hypogonadism: A state which described reduced or absence of hormone secretion by the gonads (ovaries or testes).
Hypogonadotropism: Reduced or absent gonadotropin secretion, often characterised by FSH and LH deficiency leading to testicular or ovarian dysfunction.
Hypoplasia:Incomplete development of an organ or tissue.
Nasal Fin: A plate-like ectodermal structure that forms between the medial and lateral prominences that thins to the form the oronasal membrane.
Nasal Septum: Separates the left and right airways of the nose into nostrils. It is made up of the perpendicular plate of ethmoid bone, the vomer bone, cartilage and the crest of the maxillary and palatine bones.
Olfactory bulb: The primary part of brain which processes olfactory information.
Olfactory epithelium: mucous membrane superior to the nasal cavity which contain olfactory nerve cells.
Olfactory nerve cell: Cells in the olfactory epithelium which detect various odors and signal the information to the CNS.
Olfactory Placode: A thickened area of ectoderm on the frontonasal prominence which contributes to the development of the olfactory sensory system.
Pheromone: Any molecules (scent) released by animals and affect the behavior of organisms of the same species via the olfactory system.
Vomeronasal Organ: To do with specific reproductive olfaction e.g. the detection of pheromones. A rudimentary organ in humans.
References
- ↑ <pubmed>12884838</pubmed>
- ↑ 2.0 2.1 2.2 2.3 <pubmed>16952059</pubmed>
- ↑ <pubmed>15836430</pubmed>
- ↑ <pubmed>17232381</pubmed>
- ↑ <pubmed>3548583</pubmed>
- ↑ Kollmanm, J. (1907). Atlas of the Development of Man (Vol. 2). Germany. Sourced from http://embryology.med.unsw.edu.au/embryology/index.php?title=Main_Page
- ↑ A A Pearson The Development of the Olfactory Nerve in Man J. Comp. Neurol.:1941, 75(2);199-217
- ↑ "Press Release: The 2004 Nobel Prize in Physiology or Medicine". Nobelprize.org. 27 Aug 2012 http://www.nobelprize.org/nobel_prizes/medicine/laureates/2004/press.html
- ↑ <pubmed>7143026</pubmed>
- ↑ <pubmed>17468753 </pubmed>
- ↑ <pubmed>12951145</pubmed>
- ↑ <pubmed>16269360</pubmed>
- ↑ <pubmed>11226964</pubmed>
- ↑ <pubmed>18066954</pubmed>
- ↑ <pubmed>19652915</pubmed>
- ↑ The Neurobiology of Olfaction Edited by Anna Menini. Boca Raton (FL): CRC Press; 2010. ISBN-13: 978-1-4200-7197-9
- ↑ <pubmed>19804753</pubmed>
- ↑ <pubmed>15893982</pubmed>
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 19.7 19.8 <pubmed>16093325</pubmed>
- ↑ Treloar HB, Miller AM, Ray A, et al. Development of the Olfactory System. In: Menini A, editor. The Neurobiology of Olfaction. Boca Raton (FL): CRC Press; 2010. Chapter 5. Available from: http://www.ncbi.nlm.nih.gov/books/NBK55972/
- ↑ 21.0 21.1 21.2 <pubmed>8045344</pubmed>
- ↑ <pubmed>12642481</pubmed>
- ↑ <pubmed>16677629</pubmed>
- ↑ 24.0 24.1 24.2 24.3 <pubmed>15454774</pubmed>
- ↑ <pubmed>22964415</pubmed>
- ↑ <pubmed>1281697</pubmed>
- ↑ 27.0 27.1 27.2 27.3 <pubmed>1740543</pubmed>
- ↑ 28.0 28.1 28.2 28.3 28.4 28.5 28.6 <pubmed>438867</pubmed>
- ↑ <pubmed>9712194</pubmed>
- ↑ 30.0 30.1 30.2 30.3 30.4 30.5 30.6 30.7 30.8 30.9 <pubmed>11117628</pubmed>
- ↑ <pubmed>22906231</pubmed>
- ↑ <pubmed>4069357</pubmed>
- ↑ <pubmed>5558232</pubmed>
- ↑ <pubmed>7876733</pubmed>
- ↑ 35.0 35.1 35.2 <pubmed>8544637</pubmed>
- ↑ <pubmed>881923</pubmed>
- ↑ 37.00 37.01 37.02 37.03 37.04 37.05 37.06 37.07 37.08 37.09 37.10 37.11 37.12 <pubmed>19328897</pubmed>
- ↑ Flake C.G., Ferguson C.F.: Congenital choanal atresia in infants and children. Ann Otol Rhino Laryngol 70. 1095-1110.1961
- ↑ <pubmed>7098739</pubmed>
- ↑ <pubmed> 3679682</pubmed>
- ↑ <pubmed>16155193</pubmed>
- ↑ <pubmed>3688031</pubmed>
- ↑ <pubmed>9450891</pubmed>
- ↑ <pubmed>18698631</pubmed>
- ↑ <pubmed>18698631</pubmed>
- ↑ <pubmed>11397875</pubmed>
- ↑ <pubmed>14623956</pubmed>
- ↑ 48.0 48.1 <pubmed>11232465</pubmed>
- ↑ 49.0 49.1 49.2 49.3 49.4 <pubmed>12567078</pubmed>
- ↑ 50.0 50.1 50.2 50.3 <pubmed>22882983</pubmed>
- ↑ <pubmed>6932275</pubmed>
- ↑ 52.0 52.1 52.2 52.3 <pubmed>21682876</pubmed>
- ↑ 53.0 53.1 53.2 53.3 53.4 <pubmed>12007408</pubmed>
- ↑ 54.0 54.1 54.2 54.3 54.4 54.5 <pubmed>20949504</pubmed>
- ↑ <pubmed>19021638</pubmed>
- ↑ <pubmed>1913827</pubmed>
- ↑ 57.0 57.1 57.2 57.3 57.4 <pubmed>20117945</pubmed>
- ↑ <pubmed>12627230</pubmed>
- ↑ 59.0 59.1 <pubmed>20389090</pubmed>
- ↑ 60.0 60.1 60.2 60.3 <pubmed>18826963</pubmed>
- ↑ 61.0 61.1 61.2 61.3 61.4 61.5 61.6 61.7 61.8 <pubmed>16932275</pubmed>
- ↑ <pubmed>1080088</pubmed>
- ↑ <pubmed>6881209</pubmed>
- ↑ 64.0 64.1 <pubmed>11531922</pubmed>
- ↑ <pubmed>11297579</pubmed>
- ↑ 66.0 66.1 66.2 66.3 <pubmed>11052640</pubmed>
- ↑ <pubmed>21543621</pubmed>
- ↑ <pubmed>21943152</pubmed>
- ↑ <pubmed>22906231</pubmed>
- ↑ <pubmed>22912413</pubmed>
- ↑ 71.0 71.1 71.2 71.3 <pubmed>22416012</pubmed>
- ↑ 72.0 72.1 <pubmed>22046273</pubmed>
- ↑ 73.0 73.1 73.2 <pubmed>22581782</pubmed>
External Links
Development of the Olfactory System
The Development of the Olfactory System 2
General Physiology of Olfaction
Anatomy and Physiology of Olfaction
The Kallmann's Syndrome Organisation
Image Gallery
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
--Mark Hill 12:22, 15 August 2012 (EST) Please leave the content listed below the line at the bottom of your project page.
2012 Projects: Vision | Somatosensory | Taste | Olfaction | Abnormal Vision | Hearing