2012 Group Project 3: Difference between revisions
| Line 112: | Line 112: | ||
In an animal study conducted by Rothova M, Thompson H, Lickert H, Tucker AS.(2012) exploring the historically debated issue of endoderm contribution to tongue development showed promising evidence that position of taste buds are patterned by the border of ectoderm and endoderm derivative epithelium. <ref><pubmed>ref>22581563</pubmed></ref> Research by Ozdener H, Spielman AI, Rawson NE.(2012) developing a culture which allows taste cells to survive for up to 12 months, empowers researchers to study the processes of proliferation, differentiation and function. <ref><pubmed>ref>22643728</pubmed></ref> | In an animal study conducted by Rothova M, Thompson H, Lickert H, Tucker AS.(2012) exploring the historically debated issue of endoderm contribution to tongue development showed promising evidence that position of taste buds are patterned by the border of ectoderm and endoderm derivative epithelium. <ref><pubmed>ref>22581563</pubmed></ref> Research by Ozdener H, Spielman AI, Rawson NE.(2012) developing a culture which allows taste cells to survive for up to 12 months, empowers researchers to study the processes of proliferation, differentiation and function. <ref><pubmed>ref>22643728</pubmed></ref> | ||
Brain-derived neurotrophic factor (BDNF) was identified to promote gustatory neuron development, along with identifying the embryonic precursor populations for cranial ganglia in mice through the use of fate mapping in a study by Harlow, Yang, Williams, Barlow (2011)<ref><pubmed>ref>PMC2674259</pubmed></ref> | Brain-derived neurotrophic factor (BDNF) was identified to promote gustatory neuron development, along with identifying the embryonic precursor populations for cranial ganglia in mice through the use of fate mapping in a study by Harlow, Yang, Williams, Barlow (2011)<ref><pubmed>ref>PMC2674259</pubmed></ref> | ||
The WNT gene family has a function of signally proteins for various reason such as development, In 2010 Liu, Staubach Grosse, Walton, Saims, Gumucio, Mistretta explored recent findings on the role of WNT's in tongue and papillae development, Concluding that WNT/β-catenin essential for fungiform papillae differing to WNT5a which proved to be principle in tongue development. <ref><pubmed>ref>PMC2768563</pubmed></ref> | |||
'''Mice are commonly used when using animal models in embryonic taste reasearch due to the similar embryinic development pathways, the below image shows histological stains of mice tongues''' | '''Mice are commonly used when using animal models in embryonic taste reasearch due to the similar embryinic development pathways, the below image shows histological stains of mice tongues''' | ||
Revision as of 12:10, 18 September 2012
Taste Development
Introduction to the Gustatory System
http://en.wikipedia.org/wiki/Gustatory_system
Timeline of Developmental Processes of the Gustatory System
- Sensory Taste Development; UNSW Embryology
- Embryonic and early fetal development of human taste buds: a transmission electron microscopical study [1] - Can't access full article!
- Scanning electron microscopical studies of developing gustatory papillae in humans. [2]
- Innervation of developing human taste buds. An immunohistochemical study [3]
- Evidence for stimulus access to taste cells and nerves during development: an electron microscopic study. [4]
History of Discoveries
| Date | Significant Discovery |
|---|---|
| 350BC | Aristotle writes about the basic tastes, sweet and bitter, which can be modified, he says, by salty and acidic. |
| 1901 | D. Hanig publishes a paper containing data of taste sensitivity in different regions of the tongue. The data are later misinterpreted, giving rise to the myth of the ‘tongue map’ |
| 1908 | Fifth basic taste discovered: savouriness, described as umami, which is conferred by glutamate. |
| 1931 | Bitter taste sensitivity found to vary among humans (1) |
| 1931-32 | Genetecists confirm findings about sensitivity to bitter tasting PTC and discover non-tasting is a recessive genetic trait (2,11) |
| 1939 | Geneticists show that chimpansees like humans, vary in their ability to perceive the bitterness of PTC |
| 1992 | Discovery of gustucin, a teste cell-specific G-protein, in the taste buds. Gustucin is later shown to mark bitter, umami and sweet cells (13) |
| 2000 | First taste sonsors, the T2R receptors, discovered (3) |
| 2001 | The sweet receptor is discovered (5): a combination of TaR2 and T1R3. |
| 2002 | Amino acid detector, T1R1 and T1R3 identified (6) = Umami |
| 2005 | Sweet taste receptor found (15) in the GI tract |
| 2006 | Cells for sour taste discovered, identified by PKD2L1 (4-7) |
| 2009 | The Car4 receptor, which senses the carbon dioxide in fizzy drinks is found on sour cells (8) |
| 2010 | ENaC identified as the sodium-salt taste receptor (9) |
Adult Tongue and Taste Buds – Structure and Function
The tongue is for tasting, swallowing, and speech. It is divided into an anterior two thirds and a posterior one third. These regions are divided by a V-shaped groove at the back of the tongue (sulcus terminalis). The anterior two thirds of the tongue is covered by stratified squamous epithelium, It contains roughened surface has projections called papillae that vary in shape and number. The most numerous papillae are the filiform papillae, which function to provide a surface that aids in holding food on the tongue during chewing. The larger, less numerous fungiform papillae are scattered among the filiform papillae, Occasionally taste buds may be found on fungiform papillae. Circumvallate papilae form a wide V at the sulcus terminalis. No papillae are on the posterior third of the tongue, having mucosal folds and the lingual tonsils instead. The muscles of the tongue are skeletal running in longitudinal, vertical, and transverse directions.
The Image Below is a very simplistic Hand drawn Diagram of the surface of the Tongue showing the locations of the different papillae & and also the sulcus terminalis, This image indicates the tongue from an above veiw
The functional unit of the taste bud is a taste cells, there are between 50 and 100 taste cells in each taste bud these taste cells represent all 5 different tastes. Historically is was believed that different areas of the tongue were responsible for different taste sensations although this has since been disregarded.[1]
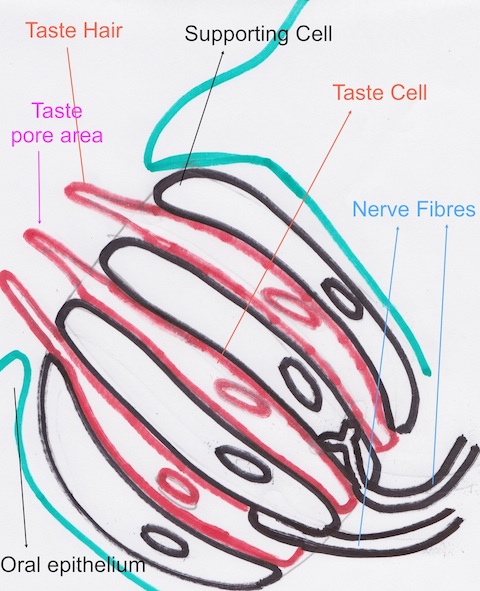
The Image Below is a very simplistic Hand drawn Diagram of a taste bud, in an extreme close up, cross section view of the the tongue
Taste Map
Abnormalities
(try to include technologies to detect abnormalities during pregnancy)
Knocking out P2X Receptors
Gustatory abnormalities has not been widely researched and what has been researched has been through animal testing, most commonly we have found on mice.
In Huang, 2008 [PMID:21940456] this team used the release of the neurotransmitter, ATP (adenosine triphosphate) as a quantitative measurement of gustatory sensation and taste. This was done by using a comparison of wild type (WT) and double knockout (DKO) mice. P2X receptors, P2X2 and P2X3 were knocked out in the DKO mice. The premise of this article was that knocking out P2X receptors reduces transmitter secretion of ATP in taste buds, therefore they cannot taste.
It should be noted that the taste buds in DKO are functional, but are not stimulated by the administration of tastants.
The transmission of release of ATP is secretion through gap junction hemichannels (pannexin 1 gap junction).
When both P2X2 and P2X3 are knocked out, no taste is elicited. However they found that if either P2X2 OR P2X3 was knocked out there was a taste response. So the inference made from this is that if one of the two receptors from the P2X family was knocked out there still can have taste response. The WT mice showed significant stimulation by tastants whereas DKO had little to no stimulation of ATP release.
Insert image 4 from article here
FGF signalling and genes
Sprouty, or Srpy, genes have been related to regulating the development of circumvillate papillae [?? check article for proper terminology] (CVM). By knocking out Spry genes using mice which had "Spry 1" and "Spry 2" knocked out showed that the number of CVP doubled. However, when "Fgf10" was absent, the number of CVP was significantly reduced, if not completely absent "'(??? check article if remember this properly).
"'Insert figure 1 from article"'
Additionally, this proves that the anterior and posterior developments of the tongue are derived from embryonic tissues, where the anterior tongue is derived fromt he ectoderm and the posterior tongue is derived from the endoderm ??? check article (& why is it necessary to know why different areas of the tongue are derived from different areas
Current Research
As time goes on we have an on going understanding of Embryonic Taste development, through the use of state of the art technology and research techniques we are able to connect the dots of this amazing natural process. The majority of research in taste development is involving mice. In an animal study using mice by Suzuki Y, Ikeda K, Kawakami K.(2011) stating that deficiencies in certain Six genes (specifically Six1 & Six4) leads to poor gustatory development. There is also evidence of a cooperative relationship between Six genes for normal development.[6] Another Animal Study involving mice explores a new idea of Neural crest contribution in taste development, specifically the development of papillae and taste buds. Liu HX, Komatsu Y, Mishina Y, Mistretta CM. (2012) suggest that Neural crest cells travel to the location of the tongue in early embryonic stages, gain epithelium phenotypes, multiply and then differentiate to eventually form taste papillae.[7] In an animal study conducted by Rothova M, Thompson H, Lickert H, Tucker AS.(2012) exploring the historically debated issue of endoderm contribution to tongue development showed promising evidence that position of taste buds are patterned by the border of ectoderm and endoderm derivative epithelium. [8] Research by Ozdener H, Spielman AI, Rawson NE.(2012) developing a culture which allows taste cells to survive for up to 12 months, empowers researchers to study the processes of proliferation, differentiation and function. [9] Brain-derived neurotrophic factor (BDNF) was identified to promote gustatory neuron development, along with identifying the embryonic precursor populations for cranial ganglia in mice through the use of fate mapping in a study by Harlow, Yang, Williams, Barlow (2011)[10] The WNT gene family has a function of signally proteins for various reason such as development, In 2010 Liu, Staubach Grosse, Walton, Saims, Gumucio, Mistretta explored recent findings on the role of WNT's in tongue and papillae development, Concluding that WNT/β-catenin essential for fungiform papillae differing to WNT5a which proved to be principle in tongue development. [11]
Mice are commonly used when using animal models in embryonic taste reasearch due to the similar embryinic development pathways, the below image shows histological stains of mice tongues
Useful links
Glossary
Image gallery
References
- ↑ <pubmed>8955790</pubmed>
- ↑ <pubmed>9455607</pubmed>
- ↑ <pubmed>9541477</pubmed>
- ↑ <pubmed>8241557</pubmed>
- ↑ <pubmed>22717400</pubmed>
- ↑ <pubmed>21978088</pubmed>
- ↑ <pubmed>22659543</pubmed>
- ↑ <pubmed>ref>22581563</pubmed>
- ↑ <pubmed>ref>22643728</pubmed>
- ↑ <pubmed>ref>PMC2674259</pubmed>
- ↑ <pubmed>ref>PMC2768563</pubmed>
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
--Mark Hill 12:22, 15 August 2012 (EST) Please leave the content listed below the line at the bottom of your project page.
2012 Projects: Vision | Somatosensory | Taste | Olfaction | Abnormal Vision | Hearing