2011 Group Project 7: Difference between revisions
| Line 161: | Line 161: | ||
Furthermore, there is a range of major and minor traits in patients with Angelman Syndrome:<ref> | Furthermore, there is a range of major and minor traits in patients with Angelman Syndrome:<ref><pubmed>16470747</pubmed></ref> | ||
'''Physical appearance''' | '''Physical appearance''' | ||
Revision as of 14:41, 6 September 2011
Angelman Syndrome
Introduction
Angelman syndrome (AS) is a rare neurogenetic disorder, first described by Dr Harry Angelman in 1956. Dr Angelman was an English paediatrician who first diagnosed the disease in 3 children with a developmental delay.
It is caused by maternal allele disruptions of a single gene-UBE3A. Either mutations or deletions of UBE3A are liable for a variety of symptoms.
AS presents with well known phenotypes during infancy and adulthood, such as microcephaly and maxillary hypoplasia, however, these features may change with advancing age due to facial coarsening. Most frequent clinical features include delayed development, seizures, motion malfunction, impairment of speech, happy demeanor, behavioural problems such as hyperactivity, short attention span and sleeping difficulty.[1] [2] [3]
The syndrome is sometimes incorrectly referred to as "happy puppet" syndrome, due to frequent laughter and excitement. [4]
About 1 in 25,000 newborn babies are affected by this disorder. There appears to be no discrepancy in males and females affected by it, and persons with the syndrome have a normal life span. [5] [6]
Up to now there is no cure for the syndrome, and current research does rather focus at improving life quality of patients with Angelman syndrome, than finding a cure. [7]
History
Timeline
1965: First reported by Dr Harry Angelman- Initially called this disorder “Happy Puppet Syndrome”
1980’s: First reports of AS reaches US and research into the disorder being at the University of Florisa under the direction of Dr. Charles Williams.
1987: Discovery of a genetic marker for AS – an absent genetic code on chromosome 15
1997: The cause of AS discovered by Dr. Joseph Wagstaff and Dr. Arthur Beaudet – mutation or deletion in the UBE3A gene.
Current: Four different genetic abnormalities for AS confirmed by genetic testing; Deletion, Uniparental Disomy (UPD), Imprinting and UBE3A mutation. [8]
| Year | Notes |
|---|---|
| 1965 | First reported by Dr Harry Angelman- Initially called this disorder “Happy Puppet Syndrome” |
| 1980s | First reports of AS reaches US and research into the disorder being at the University of Florisa under the direction of Dr. Charles Williams. |
| 1987 | Discovery of a genetic marker for AS – an absent genetic code on chromosome 15 |
| 1997 | The cause of AS discovered by Dr. Joseph Wagstaff and Dr. Arthur Beaudet – mutation or deletion in the UBE3A gene. |
| Current | Four different genetic abnormalities for AS confirmed by genetic testing; Deletion, Uniparental Disomy (UPD), Imprinting and UBE3A mutation. |
Incidence
The current population of people diagnosed with AS is unknown but studies have estimated the number to be around 1/10 000[9] and 1/20 000[10].
Aetiology
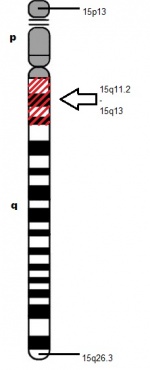
Different genetic mechanisms lead to different clinical phenotypes of AS. The most common genetic mechanism leading to AS is the deletion or re-arrangement of maternal chromosome at locus 15q11.2-q13, accounting 60-75% of AS occurrences. This leads to more severe clinical phenotypes of microcephaly, motor difficulties, seizures and impaired speech development. The next most common genetic mechanism is mutation in the UBE3A gene responsible for 10% of AS cases and paternal uniparental disomy and mutation in the imprinting centre (IC), both accounting 2-5% of AS observed.
AS is caused by the 4 major genetic mechanisms mentioned above and are thus divided into Classes I to IV based on their underlying genetic mechanism. AS patients with the clinical features of AS but no cytogenetic or molecular abnormality in Chromosome 15q11.2-13 are grouped under Class V (summarised in table below).
| Class | Mechanism | Diagnostic Tests | Frequency |
| Ia | De novo deletion | High resolution cytogenetics FISH | 60-75% |
| Ib | Deletion due to chromosome rearrangement | High resolution cytogenetics FISH | <1% |
| II | Paternal uniparental disomy | RFLP analysis | 2% |
| IIIa | Imprinting defect with IC mutation | Screening of IC for mutations is positive | 2% |
| IIIb | Imprinting defect withouth IC mutation | Screening of IC for mutations is negative | 2% |
| IIIc | Mosaic imprinting defect | Screening of IC for mutations is usually negative | ? |
| IV | UBE3A mutation | Screening of UBE3A for mutations | 5-10% |
| V | No identifiable genetic abnormality | Consider other diagnoses | 5-26% |
| Adapted from Smith JC, et al. Angelman syndrome: a review of the clinical and genetic aspects. J Med Genet 2003;40:87-95 | |||
Pathophysiology
Phenotype-Genotype correlation
Animal Models
Symptoms
Angelman syndrome causes a variety of symptoms, and the presented symptoms may vary at different ages. [11]
In 1995 Williams et al embraced the observed clinical features of AS in a consensus statement, in order to present an appliance for clinicians.
These diagnostic criteria were updated in 2005. [12]
Angelman Syndrome individuals show the most distinct disease pattern from 2 to 16 years of age. In most cases at least 8 of the symptom traits are shown. [13]
| 4 characteristics appear in 100% of the cases |
| Delayed development: becoming apparant by the age of 6 to 12 months. [14] |
| Motion and or balance malfunction |
| Behavior patterns like unmotivated laughter, frequent excitement; often including body movements, Hypermotoric behavior |
| Impairment of speech |
| 3 characteristics appear in more than 80% of the cases |
| Abnormalities in head circumference, microcephaly |
| Seizures |
| EEG shows specific pattern |
Furthermore, there is a range of major and minor traits in patients with Angelman Syndrome:[15]
Physical appearance
Behavioural appearance
Differences between patients with deletions and mutations
Diagnosis
Diagnosis of AS during early infancy is difficult as clinical features such as developmental delay, muscle hypotonia usually appear between 6 months to 2 years of age. However, unsteady or shaky movements before walking can be an early indicator of AS. Diagnosis can also be made on the basis of EGG patterns, as AS patients most typically show triphasic delta activity with a maximum over the frontal regions. The probability of this event increases with age and could help diagnose AS in mentally retarded people.
Currently two main laboratory testing methods are being used to diagnose Angelman Syndrome:
Cytogenetic approach – Is used to detect a chromosomal rearrangement or deletion at locus 15q11.2-q13 region.
Molecular testing – Involves utilising the DNA etholation test to detect a deletion, UPD (Uniparental Disomy) or an imprinting centre defect to confirm the diagnosis. This is successful in about 78% of patients. Fluorescent in situ hybridisaton studies (FISH) is also used to detect a 4-6 molecular base deletion of the 15q11.2-q13 region in about 70% of the patients by using a detecting probes such as D15S10 and/or SNRPN. The FISH test lights up or fluoresces a critical area on the chromosome 15 important for Angelman Syndrome. If there is a deletion on the chromosome, the region will not light up, indicating whether a deletion is present or not. Other molecular testing methods include sequence analysis of the UBE3A gene to detect mutations in about 10% of the patients.[16]
Differential Diagnosis
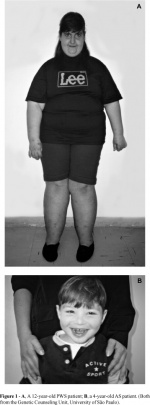
- Prader-Willi Syndrome
Results from a deletion on Chromosome 15q11.2-q13 that is inherited paternally, rather than AS that is inherited maternally.
- Rett Syndrome
Some children with the clinical diagnosis of AS but no underlying genetic mechanism show mutations in MECP2 gene, corresponding to Rett Syndrome. This should be suspected in AS test negative girls in the first few years of life.
- Mowat-Wilson Syndrome
Genetic mechanism is heterozygous deletion or truncation in ZFHX1B (SIP1) gene on 2q22. This presents with clinical features of severe intellectual disability, micrcephaly and seizures.
- X-linked alpha-thalassemia/mental retardation syndrome (ATR-X)
This arises from mutations in XNP gene on Xq13. ATR-X presents with severe intellectual disability, no speech development and seizures are common.
- Phelan-McDermid Syndrome/22q13 deletion syndrome
Deletions are usually submicroscopic and thus requires special molecular cytogenetic methods to confirm this deletion. Patients show moderate to profound intellectual disability and delay in speech development.
Prognosis
Life expectancy of AS patients seems to be normal.
Treatment and Management
| Problems encountered | Treatment and Management |
| Difficulty in feeding newborn AS babies | Use of special nipples may improve feeding |
| Gastroesophageal reflux | Special motility medications requiried or upright positioning |
| Seizures | Anticonvulsant medications |
| Ocular problems | Visual assessment is vital to encourage interactions and minimize chances of self-harm and autism development |
| Drooling in mentally retarded AS patients | Difficult to treat, but new surgical procedure on salivary duct reimplantation seems to be a promising alternative |
| Unstable children | Physical Therapy |
| AS patients become less active with increasing age | Activity schedules to prevent scoliosis and obesity. Occupational Therapy to stimulate fine motor and oral-motor control skills in conjunction to speech therapy |
| Sleeping difficulty | Sedative medication in severe cases. Melatonin may be used to promote sleeping but some studies have found that it loses its therapeutic effects after several weeks in most AS patients |
Genetic Counselling
Current and Future Research
Notable people with Angelman Syndrome
Glossary
allele: Dissimilar variant of one gene. Organisms have two alleles for each gene inherited from their parents, one from each of them.
deletion: Lack of a part of the DNA, can vary form a single base to a hole gene.
disorder: A mental or physical health disturbance or dysfunction.
mutation: Event that changes the DNA or RNA of a gene permanently.
neurogenetic: Genetic basis of the nervous system
seizure: Abnormal electrical activity in the brain that can result in a variety of physical symptoms like convulsion, body shaking, loss of consciousness, confusion, mood changes, and many more.
syndrome: Signs, symptoms or traits that appear together.
External links
http://www.angelmansyndrome.org/research.html
References
- ↑ <pubmed>21592595</pubmed>
- ↑ <pubmed>21484597</pubmed>
- ↑ http://www.angelmansyndrome.org/home.html
- ↑ http://www.ncbi.nlm.nih.gov/books/NBK22221/
- ↑ http://www.angelmansyndrome.org/home.html
- ↑ <pubmed>14510623</pubmed>
- ↑ http://www.bbc.co.uk/health/physical_health/conditions/angelman1.shtml
- ↑ http://www.angelmanproject.com/history.htm
- ↑ <pubmed>7573182</pubmed>
- ↑ <pubmed>8703225</pubmed>
- ↑ <pubmed>7625442</pubmed>
- ↑ <pubmed>16470747</pubmed>
- ↑ <pubmed>7625442</pubmed>
- ↑ <pubmed>14510623</pubmed>
- ↑ <pubmed>16470747</pubmed>
- ↑ <pubmed>19455185</pubmed>