2011 Group Project 10: Difference between revisions
m (→History) |
m (→Epidemiology) |
||
| Line 155: | Line 155: | ||
Although Duchenne Muscular Dystrophy is regarded as an X-linked recessive disorder, if often still occurs in individuals without a known family history through ''de novo'' mutations.<ref>U.S. National Library of Medicine (2011). “Genes: DMD”. Author unknown, Genetics Home Reference. Accessed via http://ghr.nlm.nih.gov/gene/DMD.</ref> | Although Duchenne Muscular Dystrophy is regarded as an X-linked recessive disorder, if often still occurs in individuals without a known family history through ''de novo'' mutations.<ref>U.S. National Library of Medicine (2011). “Genes: DMD”. Author unknown, Genetics Home Reference. Accessed via http://ghr.nlm.nih.gov/gene/DMD.</ref> | ||
== Aetiology - Genetics == | == Aetiology - Genetics == | ||
Revision as of 02:15, 10 October 2011
2011 Projects: Turner Syndrome | DiGeorge Syndrome | Klinefelter's Syndrome | Huntington's Disease | Fragile X Syndrome | Tetralogy of Fallot | Angelman Syndrome | Friedreich's Ataxia | Williams-Beuren Syndrome | Duchenne Muscular Dystrolphy | Cleft Palate and Lip
Duchenne Muscular Dystrophy (DMD)
Duchenne muscular dystrophy (DMD) is a sex-linked disorder mostly affecting males because it is a recessive X-linked disease. It is caused by a mutation in the gene that produces the important muscle protein, dystrophin. In humans this gene is located on the X chromosome, thus if a female has one affected X-chromosome then they are said to be a carrier of the disorder and can pass on the altered gene to her offspring. However, if a male inherits the altered X-chromosome they will become a sufferer of this disease because they only have one X-chromosome.
The dystrophin gene is the largest gene in nature on locus Xp21, spanning 1.5% of the X-chromosome which may explain it’s unusually high spontaneous mutation rate [1]. In DMD the protein dystrophin is not produced, when it is an important structural component for muscle tissue during contraction. Thus it results in muscle degeneration, difficulty in walking, breathing and death. The increase in muscle damage accompanies abnormal blood flow within the muscle which leads to progressive limb weakness, respiratory and cardiac failure and eventually premature death [2].
The rate of progression of the disorder is fast and the age of onset is from 2-6yrs of age.[3].
Pathologically, the main feature found in muscle biopsies from patients that suffer from DMD is fibrosis [4], the muscle is replaced with fibro-adipose tissue and it directly causes muscle dysfunction and contributes to the lethal DMD phenotype [5]. Unfortunately there is no known cure for this disorder, however due to advances in this technological era there are now many treatment methods that help delay the progress of the disease and manage the symptoms associated with it. Patients of DMD experience poor life quality and an extremely lowered life expectancy, it was only until recently, procedures that delayed the progress of the disease and that help increase the quality of life have been brought about [6].
History
Historical Background of DMD
| Date | Brief description |
| 1847 |
Dr W J Little reported two affect boys aged 12 & 14 that were unable to walk. Both were later found to have most of their leg muscles replaced with fat [7]. |
| 1851 |
Meryon described at a meeting of the Royal Medical and Chirurgical Society 8 boys presumed to be affected with Duchenne Muscular Dystrophy [7] |
| 1861 |
Duchenne first describes the disease, a young boy whose condition characterised by the hypertrophy of the boy's calf muscles [8] |
| 1868 |
Duchenne provided a much more detailed account of the boy's disease, DMD [8] |
| 1930-1960 |
Discovery of Becker Muscular Dystrophy, a less severe form form of DMD. The x-linked pattern of inheritance for DMD is confirmed.[9]. |
| 1985-1988 |
Gene responsible for DMD is found by Louis Kunkel's team & the protein made from this gene is described and named dystrophin [9] |
| 1989-1994 |
The reason for severity of DMD is found. It is determined by the amount of dystrophin present in the muscle-fibre membrane. The less dystrophin the more severe the symptoms [9] |
| 1995-2000 |
Dystrophin gene is miniturised to facilitate gene therapy and research into stem cells is on its way to cure DMD [9]. |
| 2001-2005 |
Corticosteroid prednisone trials found effective in slowing down the progression of DMD [9]. |
| 2005-present |
Important research on the autosomal homologue of dystrophin, utrophin. It is found to have a strong ameliorating effect on the pathogenesis of DMD and has extreme potential for being effective treatment for all genetic mutations of Duchenne and Becker's muscular dystrophy. For more information regarding utrophin: Important Current Research - Utrophin.
|
Dr Edward Meryon
Throughout history there have been cases that suggested muscular dystrophy, however, the first clinical descriptions of dystrophy in the English language did not appear until the 19th century. [10]. The earliest report of muscular dystrophy was from Dr Edward Meryon of St. Thomas’s Hospital, London. Born in 1809, Meryon was an English physician. He published several books concerning the nervous system and in one of his publications Meryon described eight affected boys in three families with a disease later to be identified as Duchenne Muscular Dystrophy[7]. His findings were reported in the Transactions of the Medical and Chirurgical Society in December 1851 [11]. The first systematic and detailed study of the disorder was made by Edward Meryon.
In his studies of the disease, Meryon conducted several necropsies and found the spinal cords were intact which concluded that the disease was not from the nervous system. Instead, he found that the muscles throughout the body were atrophied, soft and almost bloodless. Further microscopic examination of the muscle showed that the muscular fibres were broken down and converted into granular, fatty matter [7]. Therefore Meryon named the condition “Granular degeneration of the Voluntary muscle”. Meryon concluded there was a familiar nature to the disease that was selective for males which primarily affected muscle tissue [11].
Out of the three families he studied there were eight affected brothers and nine healthy sisters, this supported his conclusion of the disease being selective to males. Meryon’s discovery was of 10 years prior to Duchenne. He characterised the disorder as a progressive muscle wasting disease leading to premature death in the late teens, that begins in early childhood. The disease was later to be referred to as Duchenne muscular dystrophy.
Guillaume Benjamin Amand Duchenne
Guillaume Benjamin Amand Duchenne was the first to describe DMD, and thus the disease is named after him. He was born in Boulogne-sur-Mer in France, on 17 September 1806 [8]. Duchenne was a family doctor for 11 years who was interested in the study of electrical stimulation of muscle [8]. He first became interested in muscular dystrophy in 1858, and formally described it in 1861. Duchenne also invented the “harpoon” which was a needle system that he utilised to obtain percutaneous samples of muscular tissue without anesthesia [8]. This technique allowed study of material from the same patient at different stages of the disease.
Duchenne defined the disorder as:
- progressive weakness of movement first affecting the lower limb then later the upper
- pathologically; loss of striation of muscle replaced by granular matter and fat vesicles. Furthermore an increase in interstitial connective tissue in affected muscles with the production of abundant fibrous and adipose tissue in the later stages
- a gradual increase in the size of many affected muscles
- an onset during early childhood or early adolescence that is, more prevalent in boys than girls
- can affect several children in a family [12]
Epidemiology
| State | Incident rate (per 100 000 Male-liveborns) |
| Western Australia |
20.2 [13] |
| Victoria |
21.9 [13] |
| Queensland |
16.4 [13] |
| NSW & ACT |
18.6 [13] |
The incidence rate for DMD is about 1 in 3500 boys. All ethnic groups are equally affected. The most common form of muscular dystrophy found in children is Duchennes and it predominately affects males because it is an X-linked recessive disorder. Interestingly the average age of diagnosis is 5 despite the earlier onset of symptoms [14]. Between 1960 to 1971, one per 5377 liveborn males or one per 5226 liveborn males surviving to five years of age had Duchenne muscular dystrophy. Of these 64% were isolated cases meaning they were the only affected member of the family and 34% were familiar cases in New South Wales and the ACT [15].
A condition is considered X-linked if the mutated gene that causes the disorder is located on the X chromosome. Males only have one X chromosome and therefore, one altered or mutated copy of the gene is capable of causing the condition. Because of the X-linked nature of this disease in terms of its inheritance, males are more likely to develop symptoms characteristic to this disease than females. There is a high 50% chance of sons of female carriers to have the disease, with daughters having alternatively, a 50% chance of being a carrier. [16] A characteristic of X-linked inheritance is that fathers cannot pass X-linked traits to their sons.
Although Duchenne Muscular Dystrophy is regarded as an X-linked recessive disorder, if often still occurs in individuals without a known family history through de novo mutations.[17]
Aetiology - Genetics
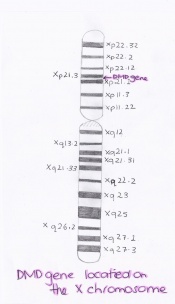
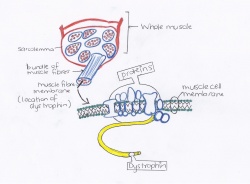
The largest gene of the human genome is called the dystrophin gene and is contained at the sacroplasmic surface of the plasma membrane or sarcolemma of muscle fibers.[18] This particular gene codes for the dystrophin protein which plays a very important role in the structural stability of muscle fibres.
The dystrophin gene, also known as DMD, is located on the short arm of the X chromosome at position 21.2. In 1987, it was found that this particular gene was identified as being located from base pair 31,137,344 to base pair 33,357,725 on the X chromosome. [19] [18] In its normal functional form, this gene produces the protein dystrophin that has multiple forms, but however is found mostly commonly in skeletal and cardiac muscles. [19]
The exact function of dystrophin has not yet been well defined, however it is suggested that dystrophin plays an important structural, protective role and signalling role during muscle contraction.
The following table summaries the 3 main roles of dystrophin in muscle fibres.
| Role of dystrophin | Brief description |
| Structural role | |
| Protective role |
|
| Signalling role | There are numerous examples that suggest a role of dystrophin in cell signalling. One example that will be given is the signalling role of alpha- syntrophin.
|
Generally, muscular dystrophies can be inherited as dominant or recessive traits, or can be due to new mutations of a specific gene. [18]As the dystrophin gene is located on the X chromosome, it can be said to be an inherited X-linked recessive condition. In the majority of affected males, the mutated gene has been inherited from the mother who is a carrier of an altered dystrophin gene, whilst a smaller minority of male cases are the result from a new mutation of this gene. In females, as they have two X chromosomes if one altered gene is expressed they are classified as carriers in that they ‘carry’ the altered gene but do not encounter any of the signs or symptoms of DMD. [20]
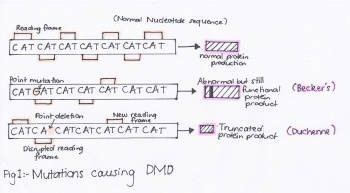
Mutations in the DMD gene can often result in the abnormal production or function of the protein, dystrophin. Some of these mutations include the deletion of part of the gene, abnormal duplication or alterations in the number of nucleotides. The two most common forms of muscular dystrophy are: Duchenne and Beckers Muscular Dystrophy.
The image based on point mutations is a visual display of the different types of mutations of the dystrophin gene that result in different forms of musuclar dystrophy. The first reading frame is that of a normal dystrophin gene and can be compared to the second reading frame that reflects a point mutation in which one of the bases has been altered, resulting in an abnormal production of dystrophin. This type of mutation results in what is clinically known as Becker’s Muscular Dystrophy (BMD). The second reading frame can then be further compared to the final reading frame that has a point deletion mutation, resulting in a new reading frame for this particular gene. The end result is a truncated protein product that is known as Duchenne Muscular Dystrophy (DMD). [21]
Pathogenesis
Dystrophin is needed in all muscle cells of the body - this includes skeletal muscles, smooth muscle and cardiac muscle. The exact function of dystrophin is unknown - it is thought to secure the sarcolemma to the actin cytoskeleton of the muscle cell. This adds strength and rigidity, protecting the muscle when it contracts[22].
In Duchenne Muscular Dystrophy, a mutation on the dystrophin gene causes a lack or absence of dystrophin, which causes many problems. Without dystrophin, the muscle cells can be easily damaged during contraction- the cell membrane becomes very permeable and allows extracellular material in. This causes swelling, until the pressure causes it to burst. Muscle fibres can also split, or begin a detrimental cycle of repeated necrosis and regeneration.[23] Necrosis often occurs in zones within the muscle fibres, a characteristic feature of Duchenne disease. The rate at which necrosis occurs is faster than the rate at which the tissue can regenerate, so the muscle fibres progressively disappear. [24].
Within the extracellular material are calcium ions, which cause serious damage when there is an influx into the muscle. Calcium activates the enzyme protease, an enzyme that breaks down proteins and peptides. In the muscle, this results in necrosis of myocytes and inflammation.[25] In the heart, increased intracellular calcium activates another protease called calpain, which deteriorates the contractile muscle[25]. This increases the stress placed on the remaining functional heart muscle.
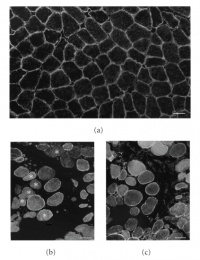
A key part of the pathogenesis is the replacement of dead muscle fibres with connective tissue (fibrosis) and adipose tissue[22]. Although components of connective tissue, such as collagen, have high tensile strength, it does not and cannot function like muscle. Significant amounts of fibroid material weaken and hinder normal muscle contraction. In the heart, this is known as cardiomyopathy, and causes serious complications for sufferers of DMD.
The picture on the right is a comparison of normal muscle tissue with DMD muscle tissue. Note the absent muscle cells, the fibrous material in between the myocytes in (b) and (c), and the lack of uniformity and rigidity. These features are characteristic of DMD.
Clinical Manifestations & Complications
Signs and Symptoms of Duchenne Muscular Dystrophy
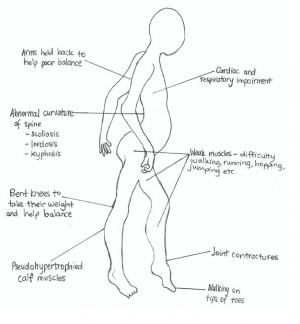
Many of the symptoms associated with DMD are due to the instability and weakness of the body's skeletal muscles. In particular, those symptoms associated with movement such as running, jumping, keeping balance and raising oneself from the ground are particularly prominent. However, he general signs and symptoms of DMD are not usually apparent until the child is 3 years old[26], or when major milestones such as learning to walk, are missed or delayed. Other complications, such as curvature of the spine or respiratory impairment are symptoms that often arise secondarily, or at a later stage in the progression of the disease. These manifestations and complications are elaborated further below.
Some typical symptoms include:
- Delayed motor movements[27]
- Frequent falls and poor balance[28]
- Weak skeletal muscles - causes difficulty running, jumping, and getting up from a sitting or lying down position[28]
- Large calf muscles (pseudohypertrophy) [28]
- Walking on toes or waddling gait[27]
- Abnormal curvature of the spine[29]
- Cardiac, respiratory and cognitive impairment[28]
Skeletal muscle
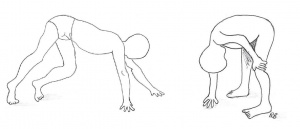
The degeneration of skeletal muscle causes many problems with mobility. In early childhood, a child affected with DMD may take longer than other children to sit or begin standing and walking. Young children may develop a waddling gait, a characteristic feature of DMD.[27] As the disease progresses, walking (especially up stairs) can become extremely difficult, and many children are confined to a wheelchair by between the ages of 8 and 11.[22] Other indicators of the disease include pseudohypertrophy (particularly of the calf muscles), fatigue, leg cramps and Gower's Sign[30]. Gower's Sign is particularly characteristic of DMD - it is where the child, from a kneeling position, will push their arms up along their legs to help them stand. A person with DMD may also suffer from joint contractures in the ankle, knees and hips[31]
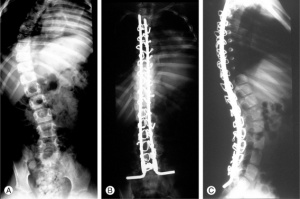
In addition to effects on body movement, DMD can cause problems with the spine. If the muscles around the spine (such as latissimus dorsi, erector spinae and trapezius muscles) weaken or atrophy, scoliosis can develop. As high as 90% of people affected by DMD will develop clinically significant scoliosis. [29] If the muscles degenerate unevenly, kyphosis[32] can occur - excessive outward curvature of the thoracic spine (resulting in a hunched or rounded back), or lordosis[33] - excessive inward curvature of the lumbar spine (resulting in a pushed forward abdomen and backwards extending hips).
Cardiac muscle
A very common and serious complication of DMD is cardiomyopathy- on average, 20% of DMD sufferers will die from cardiac failure.[22] Cardiac muscle is affected in a similar way to skeletal muscle, in which the sarcolemma loses integrity and necrotic tissue is replaced by fat and connective tissue. This severely compromises the strength and ability of the heart to contract properly and circulate blood around the body. If the heart cannot pump blood properly, cells will not receive enough oxygen for normal function. The area of the heart that is most affected is the lateral postero-basal side of the left ventricle, as this area takes the greatest strain as the heart beats[29]. Currently, there is no evidence to suggest that DMD affects the conduction system of the heart, however [34][35], in the late stages of the disease, the large quantities of fibroid material in the heart can cause systolic dysfunction and ventricular arrhythmias.
Smooth muscle
DMD in the gastrointestinal tract means the muscles cannot contract properly (peristalsis), resulting in constipation or diarrhoea. Muscles in the oesophagus can weaken, and cause difficulties swallowing food (leading to under-nutrition), pulmonary aspiration, heartburn from reflux of gastric acid[36]. Some may have delayed gastric emptying and sphincter dysfunction.[37] The stomach may lose its strength, or in the most extreme cases, patients may also suffer from acute gastric dilation or intestinal pseudo-obstruction, both of which can be fatal.[37]
Respiratory problems
Problems relating to respiratory function become most prevalent when the person requires a wheelchair or assistance in moving. By this stage of the disease, overall muscle strength is low, especially muscles such as the diaphragm and other muscles associated with breathing. The person may have difficulties breathing, or may not be able to inspire or expire to their maximum capacity. They may not be able to cough properly either.[38] As the lungs cannot function wholly, gas exchange is compromised. From this, hypercapnia may develop and can affect energy levels, weight management, cause bad headaches and disturb sleep.[29]Combined, these symptoms increase susceptibility or predispose the patient to a range of pulmonary infections, such as pneumonia. Approximately 80% of Duchenne sufferers will die from respiratory failure or a related illness.[22]
The degree of muscle strength may be (indirectly) measured by a Forced Vital Capacity (FVC) - the volume of air that can be forcibly expelled after a full inspiration. If the FVC is low, this is indicative of poor muscle strength and therefore possible respiratory failure.[29]
Cognitive Impairment
In the central nervous system (CNS), dystrophin is found:
- on the soma and proximal dendrites of pyramidal cells in the cerebral cortex and hippocampus.[39] Pyramidal cells are implicated in cognitive ability and play a role in vision-guided motor movement.[40]
- in Purkinje cells in the cerebellum[39] - Purkinje cells are large inhibitory neurons in the cortex of the cerebellum. Their main function is in controlling motor movement.[41]
The exact role of dystrophin in brain function is not known as research in this field is still in its infancy. From what has been found so far, it is believed that it affects the electrophysiology of neurons, calcium homeostasis and synaptic plasticity.[39] In particular reference to the locations above, an absence of dystrophin causes a disruption in normal brain function.
The Muscular Dystrophy Association of Australia reports that up to one third of boys will suffer from a mental disability associated with DMD. [38][22] However, very few are ‘severely’ impaired. Difficulties mostly arise in terms of emotional and social interaction – more specifically, in behavioural and communication skills. In a study of 1224 DMD patients and intelligence, it was found the average IQ was lower by one standard deviation than the normal population, and 35% of the boys had IQ scores in the mental retardation range (<70).[42] In particular, verbal skills are predominately affected, with similar learning problems to people with dysphonetic-dyseidetic dyslexia- they have difficulties with phonetics, recognising the shape of words and the synthesis of words.[42] They may also find it difficult when asked to repeat long or large pieces of information.[43]
Diagnosis
- Clinical Diagnosis - male, progressive symmetrical muscle weakness, symptoms present before age 5, elevated creatine kinase (CK) blood levels and high levels of liver enzymes (AST and ALT).[44]
- Muscle biopsy - a sample of muscle can be taken to look for abnormal levels of dystrophin in the muscle. A special stain is used to detect the dystrophin protein. In a unaffected patient, dystrophin will appear as though there is caulking around the individual muscles cells and it is holding them together like window panes. A patient suffering from DMD will have an absence of the dystrophin.
- Genetic Testing - this is achieved through a blood sample analysis. Changes in the DMD gene can be detected through various methods. E.g. Large changes in gene (deletion/duplication) or smaller components that spell out the instructions found within the DMD gene (sequencing). However, results may not be conclusive since changes in the genetic code might go undetected by the methods used.[45]
- Physical Examination - Myocardial function is an important indicator of DMD. In recent years, it has increasingly become an important cause of death.[46] Therefore, physical examination of cardiac rhythmn and function is essential in the process of diagnosing a patient with DMD. Dystrophin-deficient cardiac muscle is replaced by fibrotic tissue which can lead to wall motion abnormality, particularly in the left ventricle. These abnormalities can be detected by the following methods:[25]
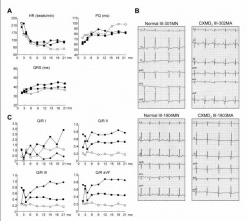
- Electrocardiography (ECG) - is able to detect myocardial scarring commonly found in DMD patients. The scarring produces sinus tachycardia. On a electrocardiograph this will appear as tall R-waves and deep Q-waves.[47] Other indicators include an increased heart rate, shortened PQ (PR) interval and arrythmias.
- Holter Monitors - monitors cardiac rhythm for a longer period of time compared to ECG and therefore can provide greater detail of sporadic abnormalities.
- Echocardiography - this method is the most universal standardised assessment of cardiac function. It uses sound waves to produce a 2D image of the heart which is clearer than an X-ray image. Echocardiography can detect myocardial thickening, wall motion abnormalities, enlargement of the left ventricle, and left ventricular systolic and dialostic function.[47]
- Cardiac magnetic resonance (CMR) - imaging is being more frequently utilized in DMD patients, providing a sensitive and reliable non-invasive measure of cardiac function.
A combination of these components along with family history confirms the diagnosis.
Current Treatments
DMD is a severe neuromuscular disease affecting male children. The progressive muscle deterioration causes the patient to become wheelchair-dependent.[48]Although there is no known cure for DMD to date, there are a variety of treatments available which are aimed at managing the symptoms, protecting muscle mass and maximising the quality of life for those who suffer from DMD. Treatments include:
| Type of Treatment | Description | Examples | Side Effects |
| Physical Therapy | Targets muscle strength and function. Research has shown that long term inactivity can weaken muscles and worsen the condition. | Regular exercise and physiotherapy sessions. Surgery may also be required in situations of severe contractures and scoliosis. | There are no side effects. |
| Orthopedic appliances | These are aimed at improving mobility and the quality of life.[49] | Braces and wheelchairs. | There are no side effects. |
| Medication | A variety of steroidal drugs are administered to treat symptoms. |
|
Weight gain, high blood pressure, behavioral changes, weakened bones and delayed growth.[45] Depression, peptic ulcers, muscle or joint pain, high blood presure, changes in vision, seizures and unusual bleeding or bruising. [51] |
Future Therapies
The following table outlines future therapies, currently being researched, that are targeted at treating and managing DMD.
| Future Therapies | Description |
| Poloxamer 188 (P188) | P188 is a non-ionic triblock copolymer, poly(ethylene oxide)80- poly(propylene oxide)27-poly(ethylene oxide)80.
Previous studies have demonstrated the beneficial capacity of P188 in preventing and reducing cardiac damage in DMD affected animals. Based on these animal studies, P188 could become an important acute therapy in DMD. Cite error: Invalid |
| Losarton | Losarton is an ATII-type1 receptor blocker which modulates ATII signaling.
Studies have shown decreased myocardial fibrosis and preservation of cardiac function in DMD mice treated with losarton over a 6 month period. Based on these findings, it is possible that losartan could decrease both skeletal and cardiac muscle fibrosis and preserve skeletal muscle strength and cardiac function in DMD patients. Clinical studies using losartan are currently in progress. [25] |
| Idebenone | Idebenone is a synthetic analog of coenzyme Q10.
It is an antioxidant medication shown to improve mitochondrial respiratory chain function and cellular energy production. A clinical trial was recently completed studying the effects of idebenone in DMD patients with cardiac dysfunction. [25] |
| Gene Therapy | Due to the lack of specific medical therapies for DMD at this time, gene therapy offers the promise of a cure by replacing the mutated dystrophin gene in all muscle tissues.[25] However this type of procedure has experienced many complications in regards to the medium of replacement and the possible side effects. |
| Stem Cell Transplant | Much of the initial focus was placed on myoblast transplantation however multiple studies showed little or no success. Research was then expanded to include stem cells that were myogenic precursors. These were obtained from bone marrow, satellite cells, muscle and blood-derived stem cells. Significant further research is required before stem cell therapy becomes a viable treatment strategy. [25] |
| Utrophin | Utrophin is an autosomal protein encoded by a gene on chromosome 6 in humans. The primary structure is very similar to that of dystrophin, being 80% identical. Current research observes the upregulation of utrophin to replace dystrophin in DMD patients. Utrophin expression is predominantly driven by two promoters: A and B. Promoter A is responsible for the skeletal muscle-specific expression of utrophin and Promoter B drives expression in endothelial cells.[53] |
Important Current Research - Utrophin
Recent research has suggested that utrophin could be highly effective in the treatment of DMD.Utrophin is the autosomal homologue of dystrophin[54]. Utrophin shares 80% similarity with dystrophin[55], with only small changes in the structure of the protein. The gene UTRN encodes utrophin and is located on band q24 of chromosome 6. This gene is approximately 1/3 of the size of the dystrophin gene. During human fetal development, utrophin is found at the sarcolemma until week 26, when it is replaced by dystrophin, suggesting that utrophin is a fetal isoform of dystrophin.[54]Utrophin expression is not affected by the DMD gene mutation, and thus could be very important for treating all DMD patients, regardless of the type of mutation[56]. Below are summaries of two important and recent papers on utrophin experiments.
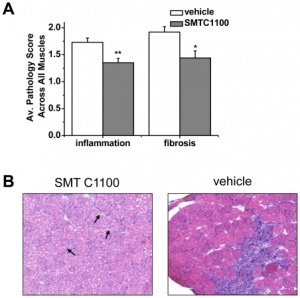
- Daily Treatment with SMTC1100, a Novel Small Molecule Utrophin Upregulator, Dramatically Reduces the Dystrophic Symptoms in the mdx Mouse
A group of researchers from England and Italy -Tinsley and Fairclough et. al, (2011)[57] developed a utrophin up-regulator and tested its effects in mdx mice. This journal describes the results of their experiment. The researchers developed an utrophin up-regulator called SMT C1100, which with daily dosing, significantly reduced the pathology and problems associated with dystrophin deficiency. In their experiment, mdx mice were grouped and treated as per table below:
| Sedentary mdx mice | Exercising mdx mice | ||||||
| Control (vehicle) | SMT C1100 | Control (vehicle) | SMT C1100 | Prednisone | SMT C1100 + Prednisone | ||
Analysis was conducted on muscle mechanics, electrophysiology, proteins, RNA, blood and histology from each group of mice. For each variable above, the researchers described in detail the effect of utrophin on the muscle cells and how it was improved. The results of the experiments showed that SMT C1100 had a significant impact in the treatment of DMD, especially when combined with Prednisone (PDN -type of glucocorticoid used to treat DMD). The increased levels of utrophin significantly reduced the dystrophy pathology of fibrosis and inflammation of the muscle cells, and led to increased strength and resistance to fatigue after exercise. Other researchers[58] have found similar results in experiments using utrophin therapies on mice.
Based on their results, the researchers argue that use of utrophin is very effective as it addresses the primary cause of dystrophy (i.e. it replaces the role of the missing dystrophin) and therefore can treat all mutations of Duchenne muscle dystrophies. They argue strongly the importance of retesting formulations of the utrophin up-regulator and its use in human DMD trials.
- Naturally occuring utrophin correlates with disease severity in Duchenne muscular dystrophy
This paper examines human DMD patients, evaluating the role of utrophin in the severity and progression of the disease. Sixteen DMD patients were investigated using muscle biopsies, muscle protein analysis, and ages at moderate disability and wheel-chair bound stage was recorded. Their results showed that DMD sufferers had up to eleven times higher levels of naturally occuring utrophin than normal adult muscle, and that utrophin expression increases with age.
Importantly, a second positive correlation was found between the quantity of utrophin at the first muscle biopsy and age at reaching wheelchair stage. From this, the authors concluded that utrophin has an ameliorating effect on muscle dystrophy and that this extended the time for which the patient could move independently. The results of this experiment are very similar to other studies[57] demonstrating the positive effect of utrophin in mice. In light of their results, the authors argue that utrophin is a suitable replacement for dystrophin, and is also a viable treatment for human muscle dystrophy disorders. They argue that further research and trials, particularly in humans, is needed.
Duchenne Muscular Dystrophy Foundations and Organisations
- Muscular Dystrophy Foundation Australia - This foundation has been set up to raise awareness of muscular dystrophy disorders and to fund research and support groups across Australia. It has offices in each state that collaborate together. They describe their values as innovative, passionate, determined and embracing.
Link to their website: http://mdaustralia.org.au/
- Muscular Dystrophy Campaign - This group is based in the United Kingdom and has a strong research and clinical trial programme. Similar to the Australian foundation, it also aims to raise funds and awareness of muscular dystrophy disorders.
Link to their website: http://www.muscular-dystrophy.org/
Glossary of terms
- Actin: a protein that forms the contractile units of muscle cells
- Acute gastric dilation: short, severe expansion or distension of the stomach, can cause the stomach to twist
- Analog: a compound that resembles another in structure
- Arrhythmias: abnormal heart contractions/irregular heart beat
- Atrophy: wasting away or disintegration of; decrease in size, owing to disease, misuse, injury
- Autosomal: a non-sex chromosome
- Base pair: two complementary bases in a nucleic acid molecule
- Cardiomyopathy: heart muscle disease
- Caulking: to make airtight or watertight by sealing or filling
- Cerebral cortex: unmyelinated neurons (grey matter) that forms the outer layer of the cerebrum
- Cytoplasm: material within a cell
- Cytoskeleton: microscopic skeleton of a cell within the cytoplasm, composed of protein
- Creatine kinase: an enzyme normally highly concentrated within muscle cells. As muscle cells degenerate, their contents are released into the bloodstream. Therefore elevated levels of creatine kinase can be detected by a blood test and is a measure of muscle damage
- Dendrites: thin extension of a nerve cell, branches out to receive nerve impulses from other neurons
- Dominant: a trait determined by an allele that is expressed over another present allele
- Dystrophin: a protein involved in muscular contraction
- Dystrophin-associated protein complex (DAPC): a multiprotein complex that includes dystrophin and other associated proteins
- Dystrophy: degenerative disorder; weakens and atrophies
- Extracellular matrix: network of connective tissues and fibres that provide support and exist outside the cell
- Fibrosis: a repair process by the body in response to injury- damaged tissue is replaced by connective tissue and often results in a scar
- Fibrotic:excess of fibrous connective tissue in an organ
- Filamentous: thread-like cells
- Gene: sequence of nucleotides forming part of a chromosome
- Genome: the complete set of genetic material in a cell or organism
- Hippocampus:sea-horse shaped mass of grey matter found on the floor of the lateral ventricles in the brain -functions in long term memory, emotion and controls the autonomic nervous system and is classified as part of the limbic system
- Homologue: a chromosome that is similar in physical attributes and genetic information to another chromosome with which it pairs during meiosis
- Hypercapnia: abnormally high levels of CO2 in the bloodstream
- Hypertrophy:enlargement of organ or tissue by increasing the size of cells
- Joint contractures: stiffness of the joints, prevents movement or full extension and flexion
- Macrophage: lymphatic cell found throughout the body; clears dead cells and debris
- Muscle contraction: process in which muscles shorter and are made tighter under neuronal control
- Muscle fiber: elongated contractile cell
- Myoblast: precursor of skeletal muscle tissue. Fusion of myoblasts gives rise to myotubes which eventually develop into skeletal muscle fibres
- Myocyte: a muscle cell
- Necrosis: cell death in a particular region of tissue
- Nucleotides: compounds that form the basic structural units of nucleic acids
- Phenotype: the physical expression of the genotype
- Plasma membrane: composed of lipids and proteins, forms the external boundary of the cytoplasm
- Protease: an enzyme that breaks down proteins and peptides
- Proximal: towards the point of attachment
- Pseudohypertrophy: enlarged muscles due to large amounts of fat and connective tissue; characteristic of DMD. Usually of the calves but may be found in other muscles such as the deltoids and serratus anterior
- Pseudo-obstruction: when a patient expresses the symptoms of intestinal blockage, but there is no physical blockage. Can be acute or chronic
- Pulmonary aspiration: entry of foreign material (food, drink, stomach contents like bile or vomit, pharyngeal secretions) from the oesophagus into the trachea or lower respiratory system.
- Recessive: the opposite of Dominant
- Sarcolemma: the thin membrane of striated muscle fibers
- Satellite Cells:
- Sacroplasmic: (sacroplasma) The cytoplasm of striated muscle cells
- Scoliosis: abnormal curvature of the spine
- Signal transduction: internal cell signalling process
- Sinus tachycardia: rhythm in which the rate of impulses arising from the SA node is elevated
- Soma:body of the neuron cell
- Systolic: maximum blood pressure during contraction of the heart
- Transmembrane: existing across a membrane
- X chromosome: sex chromosome, two are represent in females and only one is present in male cells
References
- ↑ (http://hstalks.com.wwwproxy0.library.unsw.edu.au/main/citation_info.php?c=252)
- ↑ <pubmed>21574524</pubmed>
- ↑ (http://dystrophy.com/muscular-dystrophy/Types+of+Muscular+Dystrophies)
- ↑ <pubmed>20613637</pubmed>
- ↑ <pubmed>21263136</pubmed>
- ↑ <pubmed>19774532</pubmed>
- ↑ 7.0 7.1 7.2 7.3 <pubmed>8326496</pubmed>
- ↑ 8.0 8.1 8.2 8.3 8.4 <pubmed>16225184</pubmed>
- ↑ 9.0 9.1 9.2 9.3 9.4 MDA 2011, Milestones in Duchenne Muscular Dystrophy Research, (http://www.mdausa.org/publications/milestones-embed.html)
- ↑ Alan E. H. Emery, 1987, Duchenne Muscular dystrophy, Oxford Medical Publications, New York. pp.10
- ↑ 11.0 11.1 Alan E. H. Emery, 1987, Duchenne Muscular dystrophy, Oxford Medical Publications, New York. pp.12
- ↑ <pubmed>10449553</pubmed>
- ↑ 13.0 13.1 13.2 13.3 <pubmed>7205898</pubmed>
- ↑ <pubmed> 19834452</pubmed>
- ↑ <pubmed> 7205898 </pubmed>
- ↑ Medline Plus (August, 2011). “Duchenne muscular dystrophy”. Accessed via: http://www.nlm.nih.gov/medlineplus/ency/article/000705.htm
- ↑ U.S. National Library of Medicine (2011). “Genes: DMD”. Author unknown, Genetics Home Reference. Accessed via http://ghr.nlm.nih.gov/gene/DMD.
- ↑ 18.0 18.1 18.2 18.3 18.4 18.5 Davies.K.E, Nowak. K.J (2006). "Molecular mechanisms of musclar dystrophies: old and new players." Nature Reviews. October, 2006 Volume 7, pages 763-773.
- ↑ 19.0 19.1 Genetics Home Reference. 2011. “Genes: DMD” Author anonymous. Accessed via. http://ghr.nlm.nih.gov/gene/DMD
- ↑ Genetics Home Reference. 2011. “Genetic Conditions: Duchenne and Becker muscular dystrophy.” Author anonymous. Accessed via. http://ghr.nlm.nih.gov/condition/duchenne-and-becker-muscular-dystrophy
- ↑ Medscape Reference (2011). Dystrophinopathies. Site author: Michelle L Mellion. Accessed via: http://emedicine.medscape.com/article/1173204-overview#a0104
- ↑ 22.0 22.1 22.2 22.3 22.4 22.5 Chamberlain, J. (2007), "Duchenne Muscular Dystrophy", in Dunn, B. (ed.), Protein Epidemiology: Diseases at the Level of Protein Structure and Function, The Biomedical & Life Sciences Collection, London (online at http://hstalks.com/bio).
- ↑ S Carpenter, G Karpati. Duchenne Muscular Dystrophy: Plasma Membrane Loss Initiates Muscle Cell Necrosis Unless it is Repaired. Brain: 1979, 102(1): 147-161 doi:10.1093/brain/102.1.147.
- ↑ Sarnat, H.B. (1983) Muscle Pathology and Histochemistry, American Society of Clinical Pathologists, USA: 114.
- ↑ 25.0 25.1 25.2 25.3 25.4 25.5 25.6 Spurney, C. F. (2011), Cardiomyopathy of duchenne muscular dystrophy: Current understanding and future directions. Muscle & Nerve, 44: 8–19. doi: 10.1002/mus.22097
- ↑ Bupa(2009). “Duchenne muscular dystrophy”. Accessed via: http://www.bupa.co.uk/individuals/health-information/directory/d/duchenne-muscular-dystrophy
- ↑ 27.0 27.1 27.2 http://emedicine.medscape.com/article/1173204-clinical
- ↑ 28.0 28.1 28.2 28.3 Kliegman RM, Behrman RE, Jenson HB, Stanton BF. Muscular dystrophies. In: Kliegman RM, Behrman RE, Jenson HB, Stanton BF. Nelson Textbook of Pediatrics. 18th ed. Philadelphia, Pa:Saunders Elsevier; 2007:chap 608.
- ↑ 29.0 29.1 29.2 29.3 29.4 Bushby, K., Bourke, J., Bullock, R., Eagle, M., Gibson, M. & Quinby, J. (2005) The multidisciplinary management of Duchenne muscular dystrophy. Current Paediatrics, 15, 292-300
- ↑ http://books.google.com.au/books?id=HEUZnAd4L98C&printsec=frontcover&dq=duchenne+muscular+dystrophy&hl=en&ei=xiRkTsC5B-vzmAXx8r2sCg&sa=X&oi=book_result&ct=result&resnum=1&ved=0CCsQ6AEwAA#v=onepage&q&f=false
- ↑ Stone, K., Tester, C., Howarth, A., Blakeney, J., Traynor, N., McAndrew, H., McCutcheon, M.(2007)Occupational Therapy and Duchenne Muscular Dystrophy. John Wiley & Sons, England.
- ↑ http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0002220/
- ↑ http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0003762/
- ↑ Bushby K, Muntoni F, Bourke JP. 107th ENMC international workshop: the management of cardiac involvement in muscular dystrophy and myotonic dystrophy. 7th-9th June 2002, Naarden, the Netherlands. Neuromuscul Disord 2003; 13:166-172
- ↑ Finsterer J, Stollberger C. The heart in human dystrophinopathies. Cardiology 2003; 99:1-19.
- ↑ <pubmed>7037523</pubmed>
- ↑ 37.0 37.1 <pubmed>PMID3380114</pubmed>
- ↑ 38.0 38.1 http://mdaustralia.org.au/files/2011/07/002_duchenne_becker-2011.pdf.
- ↑ 39.0 39.1 39.2 <pubmed>17720499</pubmed>
- ↑ <pubmed>18632946</pubmed>
- ↑ Rose, S. (2005) The future of the brain: the promise and perils of tomorrow's neuroscience. Oxford University Press: Oxford, England.
- ↑ 42.0 42.1 <pubmed>11463183</pubmed>
- ↑ <pubmed>18764980</pubmed>
- ↑ http://www.parentprojectmd.org/site/DocServer/Family_Guide.pdf?docID=9321
- ↑ 45.0 45.1 45.2 http://www.genome.gov/19518854#4
- ↑ Naoko Yugeta, Nobuyuki Urasawa, Yoko Fujii, Madoka Yoshimura, Katsutoshi Yuasa, Michiko R Wada et al. Cardiac involvement in Beagle-based canine X-linked muscular dystrophy in Japan (CXMDJ): electrocardiographic, echocardiographic, and morphologic studies.BMC Cardiovascular Disorders 2006, 6:47
- ↑ 47.0 47.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedyugeta - ↑ http://www.ncbi.nlm.nih.gov/pubmed/19774532
- ↑ http://jcn.sagepub.com/content/25/9/1116
- ↑ http://www.webmd.com/drugs/mono-9108-CYCLOSPORINE+CAPSULE+-+ORAL.aspx?drugid=5645&drugname=Cyclosporine+OralIt
- ↑ http://www.nlm.nih.gov/medlineplus/druginfo/meds/a601207.html#side-effects
- ↑ http://www.biomedcentral.com/1471-2261/11/20#B11
- ↑ http://ep.physoc.org/content/early/2011/09/19/expphysiol.2010.053025.long
- ↑ 54.0 54.1 <pubmed>8866746</pubmed>
- ↑ <pubmed>16595608</pubmed
- ↑ <pubmed>16443393</pubmed>
- ↑ 57.0 57.1 <pubmed>21573153</pubmed>
- ↑ <pubmed>12471059</pubmed
Kornberg, R. (2007), "Chromatin and Transcription", in Tsonis, P. (ed.), From DNA to Proteins: The Multiple Levels of Regulation, The Biomedical & Life Sciences Collection, Henry Stewart Talks Ltd, London (online at http://www.hstalks.com/bio)