2010 Lecture 3
Redirect to:
Week 1 and 2
| This is an archived lecture page. | Current page - Lecture - Week 1 and 2 Development |
Introduction
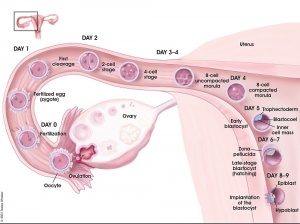
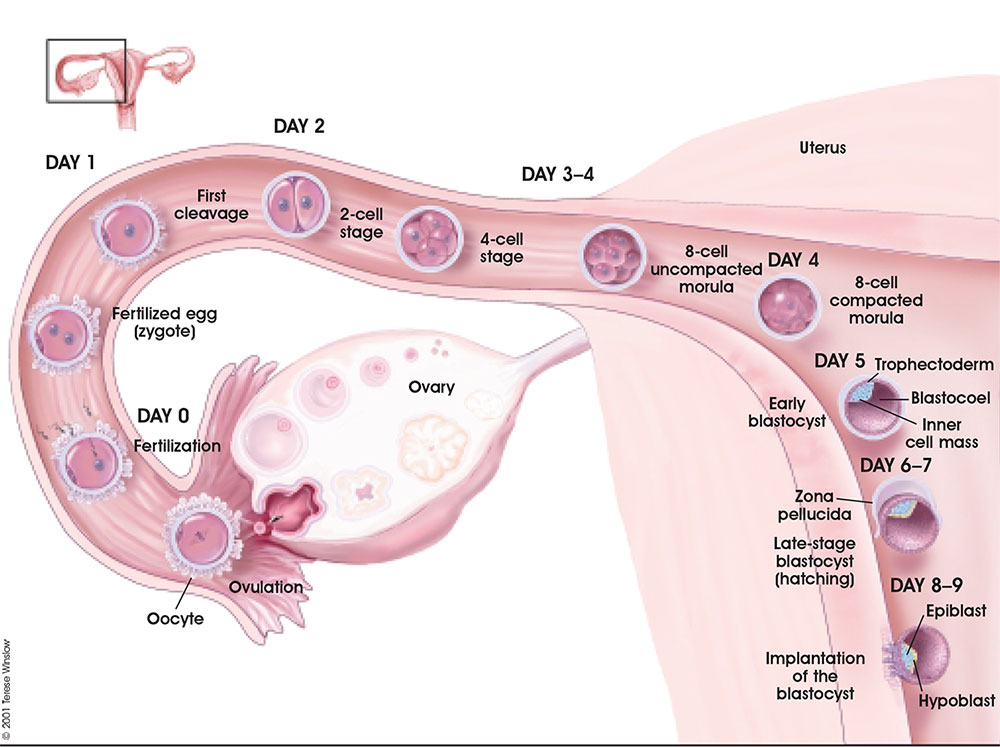
Following fertilization the first week is a series of rapid cell divisions, still contained within the zona pellucida, and still floating and being propelled within the uterine tube towards the uterine body. At the end of the first week and within the second week the process of implantation and early differentiation of cells that will form the embryo and the placenta.
| File:Week1_001 icon.jpg</wikiflv> | This animation shows the first week of human development following fertilization.
|
- Lecture Archive: 2009 Lecture | 2008 Lecture
Lecture Overview
- Week 1-2 of human development
- Fertilization options - In vitro fertilization
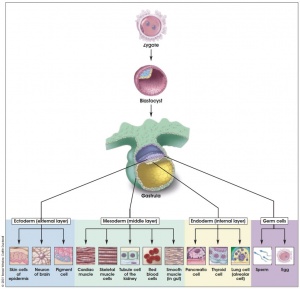
- Zygote, Morula, Blastocyst
- Cell layers - Trophoblast, Syncytiotrophoblast, Cytotrophoblast, Embryoblast
- Implantation - Normal, Ectopic, Uterine changes, Blastocyst implantation changes
Fertilization Options
- Natural sex
- Artificial insemination of mother with father's sperm, with mother with donor sperm, with egg and sperm donors, using surrogate mother
- In vitro fertilization (IVF) using egg and sperm of parents
- Intra-Cytoplasmic Sperm Injection (ICSI), frozen embryos, pre-implantation Genetic Diagnosis (PGD), egg donor, sperm donor, egg and sperm donor, surrogate using parents' egg and sperm, surrogate and egg donor, surrogate and sperm donor, surrogate using her egg and sperm from baby's father, surrogate using egg and sperm donors
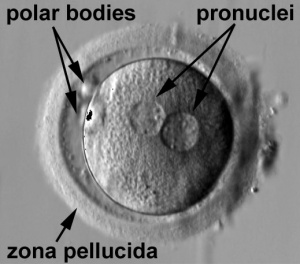
Week 1 - Formation of Zygote
- male and female pronuclei, 2 nuclei approach each other and nuclear membranes break down
- DNA replicates, first mitotic division
- sperm contributes centriole which organizes mitotic spindle
Movie - Pronuclear Fusion | Movie - Parental Genomes
Conceptus - term refers to all material derived from this fertilized zygote and includes both the embryo and the non-embryonic tissues (placenta, fetal membranes).
Cleavage of Zygote
Movie - Early Cell Division | Movie - Week 1 Cell Cleavage | Carnegie stage 1
- cleavage of zygote forms 2 blastomeres and is cleavage with no cytoplasm synthesis
- special "embryonic" cell cycle S phases and M phases alternate without any intervening G1 or G2 phases (MSMSMSMS, adult MG1SG2) therefore individual cell volume decreases
- cell division is initially synchronous, then asynchronously
- slow- centre cells, larger fast- peripheral cells
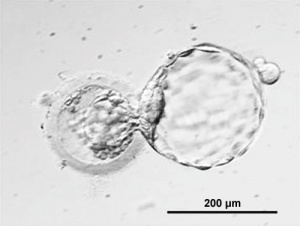
Morula
- about day 4 is a solid ball of 16-20 cells with peripheral cells flattened against zona pellucida
- compaction occurs forming a cavity and leading to the next blastocyst stage
Blastocyst
- about day 5 have 2 identifiable cell types and a fluid-filled cavity (blastoceol)
- trophoblast layer - peripheral flattened cells, forms the placenta and placental membranes
- inner cell mass - embryoblast, mass of rounder cells located on one wall of the blastocoel, forms entire embryo
Blastula Cell Communication
Two forms of cellular junctions Figure 21-69. The blastula
- gap junctions, allow electrically couple cells of epithelium surrounding a fluid-filled cavity
- tight junctions, close to outer surface create a seal, isolates interior of embryo from external medium
Blastocyst Hatching - zona pellucida lost, ZP has sperm entry site, and entire ZP broken down by uterine secretions and possibly blastula secretions. Uterine Glands - secretions required for blastocyst motility and nutrition
Week 2 - Implantation
The second week of human development is concerned with the process of implantation and the differentiation of the blastocyst into early embryonic and placental forming structures.
- implantation commences about day 6 to 7
- Adplantation - begins with initial adhesion to the uterine epithelium
- blastocyst then slows in motility, "rolls" on surface, aligns with the inner cell mass closest to the epithelium and stops
- Implantation - migration of the blastocyst into the uterine epithelium, process complete by about day 9
- coagulation plug - left where the blastocyst has entered the uterine wall day 12
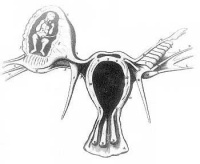
Normal Implantation Sites - in uterine wall superior, posterior, lateral
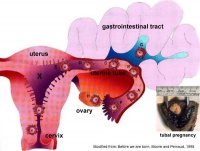
Abnormal Implantation
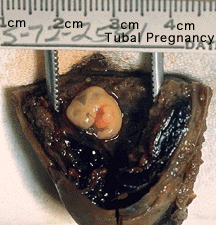
Abnormal implantation sites or Ectopic Pregnancy occurs if implantation is in uterine tube or outside the uterus.
- sites - external surface of uterus, ovary, bowel, gastrointestinal tract, mesentry, peritoneal wall
- If not spontaneous then, embryo has to be removed surgically
Tubal pregnancy - 94% of ectopic pregnancies
- if uterine epithelium is damaged (scarring, pelvic inflammatory disease)
- if zona pellucida is lost too early, allows premature tubal implantation
- embryo may develop through early stages, can erode through the uterine horn and reattach within the peritoneal cavity
|
|
Uterus
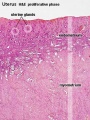
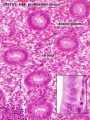
- Endometrium - 3 layers in secretory phase of menstrual cycle: compact, spongy, basal
- Myometrium - muscular layer outside endometrium, contracts in parturition
- Perimetrium - tunica serosa of the uterus continuous with the peritoneal wall
Endometrial Layers
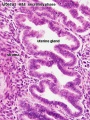
- Compact - implantation occurs in this layer, dense stromal cells, uterine gland necks, capillaries of spiral arteries
- Spongy - swollen stromal cells, uterine gland bodies, spiral arteries
- Basal - not lost during menstruation or childbirth, own blood supply
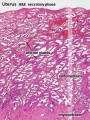
Decidual Reaction
- occurs initially at site of implantation and includes both cellular and matrix changes
- reaction spreads throughout entire uterus, not at cervix
- deposition of fibrinoid and glycogen and epithelial plaque formation (at anchoring villi)
- presence of decidual cells are indicative of pregnancy
Cervix - at mouth of uterus, secretes mucus (CMP), forms a plug/barrier, mechanical and antibacterial Vascular - increased number of blood vessels
Decidua
The endometrium becomes the decidua and forms 3 distinct anatomical regions (at approx 3 weeks)
- Decidua Basalis at implantation site
- Decidua Capsularis enclosing the conceptus
- Decidua Parietalis the remainder of uterus
- Decidua Capsularis and Parietalis fuse eventually fuse and uterine cavity is lost by 12 weeks
Uterus Abnormalities
Endometriosis endometrial tissue located in other regions of the uterus or other tissues. This misplaced tissue develops into growths or lesions which respond to the menstrual cycle hormonal changes in the same way that the tissue of the uterine lining does; each month the tissue builds up, breaks down, and sheds. Endometriosis
Bilaminar Embryo
Movie - Week 2 Bilaminar Embryo
- about day 8 to 9
The outer trophoblast and inner embryoblast layers now both differentiate to form two distinct cellular layers. The trophoblast layer forms the syncitotrophoblast and cytotrophoblast layers. The embryoblast (inner cell mass) forms the epiblast and hypoblast layers. This early stage of embryo development is referred to as the bilaminar embryo.
Syncitiotrophoblasts
- secrete proteolytic enzymes, enzymes break down extracellular matrix around cells
- Allow passage of blastocyst into endometrial wall, totally surround the blastocyst
- generate spaces that fill with maternal blood- lacunae
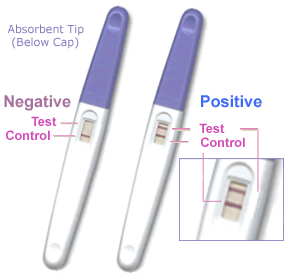
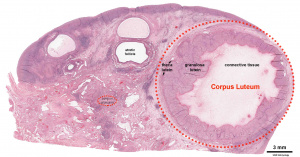
- secrete Human Chorionic Gonadotropin (hCG), hormone, maintains decidua and Corpus Luteum, basis of pregnancy diagnostic test, present in urine is diagnostic of pregnancy
- Later in development placenta will secrete hCG
MH - more information in lecture 4 and laboratory.
Human Chorionic Gonadotropin
- levels peak at 8 to 10 weeks of pregnancy, then decline and are lower for rest of pregnancy
- 0-1 week: 0-50 mIU/ml
- 1-2 weeks: 40-300 mIU/ml
- 3-4 weeks: 500-6,000 mIU/ml
- 1-2 months: 5,000-200,000 mIU/ml
- 2-3 months: 10,000-100,000 mIU/ml
- 2nd trimester: 3,000-50,000 mIU/ml
- 3rd trimester: 1,000-50,000 mIU/ml
Non-pregnant females: <5.0 mIU/ml Postmenopausal females: <9.5 mIU/ml
Twinning
Twinning can be due to two separate fertilization events (dizygotic twins) or as an abnormality of a single fertilization (monozygotic twins) event during the early weeks of development.
Dizygotic Twinning
Dizygotic twins (fraternal, non-identical) arise from separate fertilization events involving two separate oocyte (egg, ova) and spermatozoa (sperm).
- In dizygotic twinning the genetic material is different and implantation and placentation is also different.
Monoygotic Twinning
- In monozygotic twinning the genetic material is initially identical and degree of twinning will depend upon the timing (early to late) from separate fetal membranes and placenta to conjoined twins.
- morula stage (diamniotic dichorionic), early blastocyst (diamniotic monochorionic), late blastocyst to bilaminar (monoamniotic monochorionic), bilaminar to trilaminar embryo (conjoined)
- Monozygotic twins are a unique research resource for comparing environmental effects on development and health.
- Congenital abnormality statistics for twins is generally increased in various conditions.
Monoygotic twins (identical) produced from a single fertilization event (one fertilised egg and a single spermatazoa, form a single zygote), these twins therefore share the same genetic makeup. Occurs in approximately 3-5 per 1000 pregnancies, more commonly with aged mothers. The later the twinning event, the less common are initially separate placental membranes and finally resulting in conjoined twins.
Table based upon recent Twinning Review.[1]
- Links: Twinning | Australian Twin Registry
Week 1 Human Development Overview
Movies
| Ovulation | Fertilization | Pronuclear Fusion | Week 1 |
Glossary Links
A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers
References
- ↑ <pubmed>12957099</pubmed>
Textbooks
- The Developing Human: Clinically Oriented Embryology (8th Edition) by Keith L. Moore and T.V.N Persaud - Chapter 2
- Larsen’s Human Embryology by GC. Schoenwolf, SB. Bleyl, PR. Brauer and PH. Francis-West - Chapter 2
Online Textbooks
- Developmental Biology by Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000 Figure 11.22. The cleavage of a single mouse embryo in vitro | Figure 11.25. Mouse blastocyst hatching from the zona pellucida | Figure 11.20. Development of a human embryo from fertilization to implantation | Figure 11.24. Implantation of the mammalian blastocyst into the uterus
- Molecular Biology of the Cell 4th ed. Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter New York and London: Garland Science; c2002 - Fertilization | Figure 21-84. Scanning electron micrographs of the early mouse embryo | Figure 21-69. The blastula
- Molecular Cell Biology by Lodish, Harvey; Berk, Arnold; Zipursky, S. Lawrence; Matsudaira, Paul; Baltimore, David; Darnell, James E. New York: W. H. Freeman & Co.; c1999 Chapter 13. Regulation of the Eukaryotic Cell Cycle
- The Cell - A Molecular Approach by Cooper, Geoffrey M. Sunderland (MA): Sinauer Associates, Inc.; c2000 Figure 14.37. Meiosis of vertebrate oocytes
- HSTAT - In Vitro Fertilization As A Medical Treatment For Male or Female Infertility
- Human Molecular Genetics 2 Strachan, Tom and Read, Andrew P. New York and London: Garland Science; c1999 Figure 8.19. Changes in DNA methylation during mammalian development
Search
- Bookshelf blastocyst | morula | implantation | ectopic pregnancy
- Pubmed blastocyst | implantation | ectopic pregnancy |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Course Content 2010
Embryology Introduction | Cell Division/Fertilization | Lab 1 | Week 1&2 Development | Week 3 Development | Lab 2 | Mesoderm Development | Ectoderm, Early Neural, Neural Crest | Lab 3 | Early Vascular Development | Placenta | Lab 4 | Endoderm, Early Gastrointestinal | Respiratory Development | Lab 5 | Head Development | Neural Crest Development | Lab 6 | Musculoskeletal Development | Limb Development | Lab 7 | Kidney | Genital | Lab 8 | Sensory | Stem Cells | Stem Cells | Endocrine | Lab 10 | Late Vascular Development | Integumentary | Lab 11 | Birth, Postnatal | Revision | Lab 12 | Lecture Audio | Course Timetable
Cite this page: Hill, M.A. (2024, April 19) Embryology 2010 Lecture 3. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2010_Lecture_3
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G