2010 Group Project 6
Maternal serum alpha-fetoprotein
Introduction
Maternal Serum Alpha Fetoprotein (MSAFP) screening is an non-invasive procedure in which the mother’s blood is taken and alpha-fetoprotein levels are measured. It is usually carried out during the 2nd trimester and is used to detect abnormalities such as neural tube defects, more specifically anencephaly spina bifida,encephalocele, open ventral wall defects such as gastroschisis as well as Down’s Syndrome. [1]
AFP was first discovered almost half a century ago in 1963 by Gary Israilevich Abelev and much research was carried out in the 1970’s which uncovered a link between AFP levels in women during pregnancy and the onset of anencephaly and spina bifida. Coming off from this, there was a steady decline in the number of cases of anencephaly and spina bifida in the United States. Surveillance of the birth defects in the Unites States show that there were significant reductions in birth defects from 1985 – 1994. Data from other countries such as England, France and Scotland show a marked decrease in birth defects during the mid 1980’s as well. However, with the availability of other diagnostic tools such as ultrasound, amniocentesis and chorionic villus sampling, it seems that AFP screening has taken more of a secondary role – in Australia at least – in terms of commonly used screening/diagnostic tests used nowadays.
A video interview with Gary Abelev can be found on youtube if you click here.
What is Alpha fetoprotein
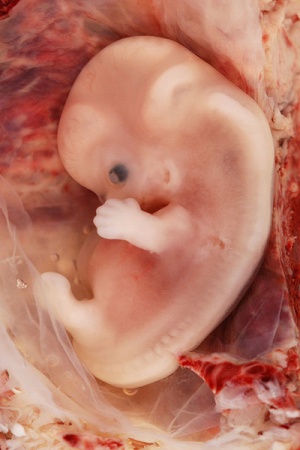
Alpha-Fetoprotein (AFP) is an embryo specific glycoprotein which is produced during the early stages of development by the liver, yolk sac as well as a small amount being produced by the gastrointestinal tract. AFP in adults is functionless as levels decrease drastically after birth with very low traces of AFP found in the average older adult with the only women experiencing spikes occurring in AFP levels during the onset of pregnancy and it is in fact through the testing of the blood of pregnant women, that AFP levels can be measured. The function of AFP itself is unknown but due to its similarity to albumin[2] it has been hypothesized that AFP could be a carrier protein or may even play a role in the metabolism of bilirubin or even may play a role in the control of female fertility through its anti-estrogenic actions[3]. Furthermore, it has been observed that it does play a role in the embryonic and early fetal stages of development as fluctuating levels of AFP indicate the presence of abnormalities within a fetus.
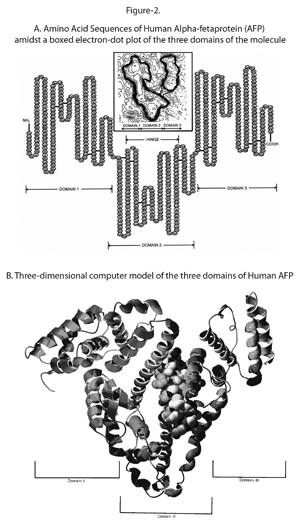
AFP has a molecular weight of around 70,000 daltons and is a single chain alpha globulin that has 590 amino acids and is estimated to have a 5% make up of carbohydrate content. The AFP level in human fetal serum is highest during the 13th week of gestation, where it may reach the level of several mg per ml, and accounts for almost a third of the total serum protein. Normal human serum also contains traces of AFP, however fetal AFP level is almost one million times higher than the adult level.
Ranges and Levels
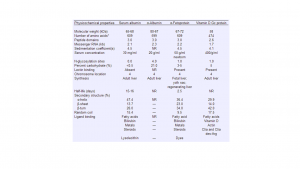
AFP blood test ranges will vary between groups of people when factors such as age and sex come into play. However, a general trend for normal AFP levels in people is as follows:
| Men | 0-20 ng/mL |
| Women | 0-20 ng/mL |
| Women (Pregnant) | Ranges can be separated into First Trimester and Second Trimester Results as presented below |
First Trimester
- 200 - 400 mg/dL
Second Trimester
- 14 weeks of gestation: 25.6 ng/mL
- 15 weeks of gestation: 29.9 ng/mL
- 16 weeks of gestation: 34.8 ng/mL
- 17 weeks of gestation: 40.6 ng/mL
- 18 weeks of gestation: 47.3 ng/mL
- 19 weeks of gestation: 55.1 ng/mL
- 20 weeks of gestation: 64.3ng/mL
- 21 weeks of gestation: 74.9 ng/mL
It should also be noted that 'normal' values are around 200% higher is women with twin pregnancies. Furthermore, it was found that the 'normal' value of AFP was 15% higher in African Americans when compared to Caucasians. [4]
AFP in Pregnancy
Highest maternal AFP concentration occurs in the mid third trimester of the pregnancy where the mean level is 150-250ng/ml. The concentration of AFP in maternal serum at any moment of gestation development seems to be related to the AFP level in the fetal circulation as well as in the placental size.
Instances of abnormal AFP values (too high as well as too low[5]) can partly been explained by physiological deviations from the expected normal pregnancy eg. in cases of under- or overestimated gestational age and multiple pregnancies. In other instances it have been found to indicate the presence of various fetal morphogenetic defects, such as open NTD (neural tube defect), hereditary congenital nephrosis (Finnish type), omphalocele, pilonidal sinus, esophageal atresia, and others.
The maternal AFP level has often reported to be increased in pregnancies where the fetus has a neural tube defect.
The Optimal practical time for detecting open spinabifida by measuring materal serum AFP is at 16-18 comepleted weeks of pregnancy. In Wald et el. (1977)’s sample of patients, 88% of cases of anencephaly, 79% of cases of open spina bifida, and 3% of unaffected singleton pregnancies had maternal serum AFP levels equal to or greater than 2.5 times the normal median. Because there is a certain degree of overlapping between the maternal AFP levels in pregnancies with and without fetal NTD, the AFP estimation in materal serum cannot per se serve as a specific diagnostic test, but it seems to be a useful screening test so as to select certain symptom-free women for further diagnostic procedures such as ultrasonography, amniocentesis, and amniography[6].
Maternal Serum Alpha Protein as a Screening Test
It should be made clear that MAFP is not a diagnostic test and is used only for screening purposes to determine the likelihood of a disease being present, with further testing always necessary for any sort of accurate diagnosis to take place[7]. Furthermore, MAFP is a screening test that is carried out during the second trimester whereas other tests may be carried out during the first trimester and are more accurate. It is part of two tests, one called the Triple Screen Test which is a battery of tests that measure AFP levels as well as human chorionic gonadotropin (hCG) and unconjugated estriol uE3 and a second series of tests known as the Quadruple Screen Test[8] that tests AFP, hCG, uE3 as well as Inhibin A which is a hormone that is released by the placenta. These tests also take into account age, ethnic background, weight as well as the babys' gestational age. Currently, there are no known risks or side effects that have been associated with the MSAFP screening test except for any discomfort involved with the drawing of blood from the patient.
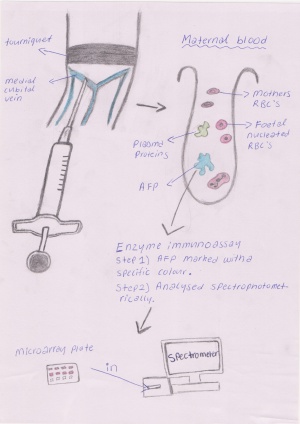
When the maternal blood serum is being tested and has been collected the alpha fetoprotein undergoes an enzyme immunoassay procedure in order to determine the concentration of the protein in the blood. Firstly AFP is marked, usually with a colour or florescence marker, then the assay is placed in a spectrometer for a result on the concentration. Other less common procedures which are used to determine the concentration of AFP are radio-immuno assay, bioluminescence and chemiluminescence methods.[9]
Previously, the use of MSAFP as a screening test was called into question in regards to its accuracy as well as its cost effectiveness as a medical program from the perspective of a managed health care system (note that this was from the view of an American health insurer). It was concluded that MSAFP would not result in a cost savings to the insurer however, it would be cost-justified when viewed from the perspective of society when other reasonable assumptions where taken into account. In Australia, the MSAFP screening test isn't as commonly used as other first trimester tests however, it is one of the few pre-natal tests that is covered by medicare whereas all the first trimester tests available are payed by the patients themselves[10].
MSAFP Screening Test Procedure
The procedure requires blood to be drawn from the patient and there are two methods that can be used - venous or umbilical blood sampling:
- Venous blood sampling, a needle is usually inserted into the vein in your arm and blood will be collected into a tube.
- Umbilical blood sampling is called percutaneous umbilical blood sampling and a needle is inserted into the mother's abdomen and into the umbilical cord. This procedure has a few more associated risks than the standard venous blood sampling procedure as there are chances, albeit extremely low, that there may be bleeeding from the puncture site, heart rate of the baby being affected - fetal bradycardia, infection or even thrombosis of the umbilical vein.
Advantages and Disadvantages of MSAFP
Advantages:
- In Australia, the MSAFP test is covered by medicare, thus, it is a financially viable test
- When used as part of the Triple or Quadruple Tests, MSAFP is a non-invasive screening test
Disadvantages:
- The accuracy of MSAFP screening is not as reliable as other pre-natal diagnostic tests due to the presence of false-positive results. The real danger, is the follow up of an invasive diagnostic test such as amniocentesis or chorionic villus sampling which have a 1 - 2% rate of fetal loss
- MSAFP can only be performed during the 2nd trimester between weeks 15 - 20
Accuracy of the MSAFP Sceening Test
The accuracy of MSFAP has always been a controversial issue with around a claim of a 5% false-positive rate, however more recent data suggests that around 80% of positive tests where the baby is in actual fact unaffected by any abnormalities that may have been expressed. Taking into account this discrepancy of results, the standard procedure is to repeat the MSAFP test and following a second positive result, ultrasound and/or amniocentesis is used.
Disorders that MSAFP indicates
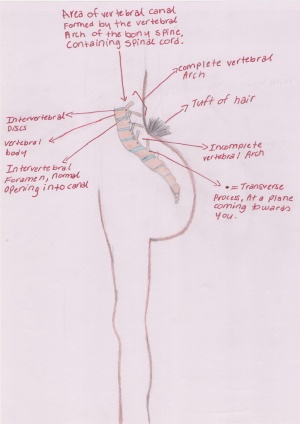
Spina bifida and Anencephaly
MSAFP level testing is good for detection for spinal bifida and anencephaly. While ultrasonic examination is capable of diagnosing anencephaly in utero, it is unlikely to be widely available as a screening procedure for all pregnant women, and there is no satisfactory way of diagnosing spina bifida in early pregnancy. AFP estimations can be performed early in pregnancies without the knowledge of the outcomes of the pregnancies. In Wald’s study (1974)[11] it was found that the pregnancies which turn out to have either spina bifida or anencephaly have a higher level of MSAFP than those in the control pregnancies matched for maternal age, parity, and length of gestation. Even though it is impossible to say with complete confidence that a fetus is unaffected if the MSAFP did not rise above normal levels, by measuring the maternal serum AFP levels we can say with a defined degree of confidence the likelihood of a pregnancy leading to spina bifida or anencephaly[12].
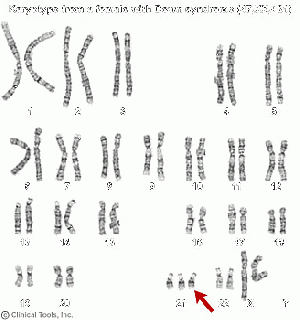
Down Syndrome
MSAFP levels are also sufficient to form the basis of a screening test for fetus with Down syndrome as they are significantly lower in pregnancies associated with Down syndrome than in unaffected pregnancies. Using a MSAFP cut-off level of 0.5 multiples of median at 14-20 weeks of gestation, excluding any of these that ultrasound cephalometry shows to have been due to overestimation of gestational age, Cuckle (1984)[13] identified 21% of pregnancies with Down syndrome as well as 5% of unaffected pregnancies.
If amniocentesis were offered to all women aged 38 or above and to younger women with serum AFP below specific maternal age-dependent cut-off levels the percentage would increase to 40% for picking up pregnancies with Down syndrome and 6.8% unaffected pregnancies.
The low AFP levels in pregnancies with Down syndrome cannot be explained by known factors associated with low AFP (i.e. maternal weight, birth weight, fetal sex, maternal diabetes mellitus). However it suggests that less AFP is produced by the fetal liver (being the main source of AFP at this time of the pregnancy) than in unaffected pregnancies.
Outside of pregnancy, the MSAFP test may be performed as part of a routine health screening especially if there is the potential of the presence of a disease or toxicity such as a liver carcinoma, or testicular cancer.
Other instances where maternal serum AFP levels are elevated includes:
- Fetus effected by hereditary cogenital nephrosis of the Finnish type - an hereditary, autosomal recessive disease which leads to death in early infancy.
- Meckel syndrome early enough in gestation to permit termination
- Intrauterine death
- Multiple gestations such as twin pregnancies or triplets
In conclusion, aberrant AFP values in maternal serum samples are to be regarded as unspecific warning signals, which sometimes may be observed weeks in advance of any other clinical or biochemical symptom of a deviant fetal development. Therefore, more specific diagnostic measures must be employed to verify and characterize the type of pregnancy disturbance that may exist. Nevertheless, the determination of AFP in maternal serum provides valuable information concerning the progress of pregnancy. All pregnant women having had a neural tube defect fetus before should be offered determination of amniotic fluid AFP at about the 16th week of gestation.[14]
MSAFP testing and the community
Ethical issues are also a factor that must be looked at when evaluating AFP testing. It is widely known that religious groups have taken strong stands against bioethical issues of stem cell research, cloning and abortions. This is a relevant issue concerning AFP testing as when parents of unborn children carry out the prenatal test and have found problems, including downs syndrome, termination or abortions are often carried out, studies have shown that in some cases an abortion was carried out 72 hours after finding a problem from a prenatal test. This is carried out due to parents deciding to abort and doctors trying to avoid ‘late’ gestation abortions complications. This raises the issue of prenatal testing as this ‘quick’ decision may be carried out due to the shock realization that their child may be born with a birth defect that cannot be reversed leading to future problems[15].
Iles and Gath found that nearly one half of the women in their study had symptoms of grief six months after the abortion and almost one third continued to grieve thirteen months after the termination. Studies also showed that unplanned pregnancy abortions had the same mental effects as planned pregnancy abortions due to parents developing maternal attachments. Therein lies the problem associated with Alpha feta protein testing as it may cause parents to abort after learning of defects with the fetus.
Issues that arise from prenatal testing can also have effects on the community at large. The contrast of termination and prenatal testing shows that women are less inclined to get tested because they are concerned about how they would feel if they found genetic disorders with their unborn baby. The grief that other parents have endured causes others to not conduct prenatal tests including AFP testing. Hvidovre University Hospital conducted research into the likelihood of women declining this test and why they did so. The most interesting finding of the survey was that of women who had a previous spontaneous abortion 24.1% refused to test while 14.8% of women who didn’t, accepted to do the test. This shows that women are refusing the test because they are aware they may get an abortion if the fetus has a defect and are conscious of the grief and guilt that may follow an abortion. The report also showed that women who were against abortions were less inclined to have the test further proving the previous point[16][17].
These findings have shown that women are aware of the problems that prenatal testing may cause and are choosing denial over truth. This has caused concern in the medical field because refusing the test may lead to children being born with birth defects unexpectedly or with problems that can be corrected inside the uterus going unresolved. It is important for the ALF test to be used even if ethical issues arise from it because knowing of any problems will allow treatment or proper management to be used which is important in ensuring unexpected problems don’t occur.
Glossary
AFP: Produced in the yolk sack during early pregnancy and then developed in the liver later in pregnancy. Function is unknown as it is very similar to albumin. Test of its concentration used to detect high or low levels to indicate possible birth defects.
Albumin: A protein produced in human liver and is tested for concentration to indicate diseases in liver of kidneys. The test shows if the body is absorbing correct amounts of protein.
Amniocentesis: A diagnostic test in prenatal circumstance where amniotic fluid is taken from the amniotic sack that contains fetal tissue. This test is used to find chromosomal abnormalities and fetal infections. Disorders found include downs syndrome.
Amniography: A procedure used to detect placement of the placenta by x-ray examination with injection of a radiopaque contrast medium into the amniotic fluid.
Anencephaly: Neural tube defect where the neural tube fail to close completely and the crianal end of the tube.
Bilirubin: The pigment of bile that is produced in the liver. Tested for amounts in blood to indicate disease like jaundice.
Carrier protein: Transport specific protein that helps substances move across interstitual spaces or cell membranes that cannot move on their own.
Downs syndrome: A chromosomal disorder where there is a 21st chromosome in the fetus. This leads to problems in growth and cognitive ability.
Gestation: Development of an embryo, approximately 9 months for humans.
Glycoprotein: A compound in which carbohydrate is covalently linked to protein. They occur in cells, in both soluble and membrane-bound forms, as well as in the intercellular matrix and in extracellular fluids, and include numerous biologically active macromolecules.
Immunoassay is a biochemical test that measures the presence or concentration of a substance in solutions that frequently contain a complex mixture of substances.
NTD (neural tube defect): Problem that occurs early in pregnancy, occurs when flat region of the spinal cord doesn’t close up during folding.
Omphalocele: Defect occurs when small intestines form outside the fetal abdomen and fail to enter the abdomen before birth.
Spina Bifida: Birth defect caused by the incomplete closure of the neural tube. This causes vertebra in the fetus to no fuse.
Utrasonography: A diagnostic test used to visualize subcutaneous structures in a body or a fetus including joints muscles and tendons. This checks for defects or problems associated with these structures.
Links
Alpha Fetoprotein [1]
Spina Bifida [2]
Down Syndrome [3]
References
- ↑ <pubmed>7534926</pubmed>
- ↑ G J Mizejewski Mapping of Structure-Function Peptide Sites on the Human Alpha-fetoprotein Amino Acid Sequence, Atlas Genet Cytogenet Oncol Haematol (2009) http://atlasgeneticsoncology.org/Deep/MappingAFPID20077.html
- ↑ <pubmed> PMC2716789</pubmed>
- ↑ Alpha-1-fetoprotein measurement, serum (2010). https://ssl.adam.com/content.aspx?productId=49&pid=49&gid=150027&site=welldynerx.adam.com&login=well1815
- ↑ The Serum Alpha-Fetoprotein Blood Test: Screening for Birth Defects (2009) http://www.brighthub.com/science/medical/articles/30994.aspx
- ↑ <pubmed> 69055</pubmed>
- ↑ Maternal Serum Alpha-Fetoprotein Screening (MSAFP) (2006) http://www.americanpregnancy.org/prenataltesting/afp.html
- ↑ Quadruple Screen Test (2010) http://www.nlm.nih.gov/medlineplus/ency/article/007311.htm
- ↑ Van Xu, H. Brian Haisail,1 and William R. Helneman, Heterogeneous Enzyme Immunoassay of Alpha-Fetoprotein in Maternal Serum by Flow-Injection Amperometric Detection of 4-Aminophenol. http://www.ncbi.nlm.nih.gov/pubmed/1700742
- ↑ S H Taplin, R S Thompson, D A Conrad Cost-Justification Analysis of Prenatal Maternal Serum Alpha-feto Protein Screening, Medical Care: 1988, 26(10); 1185-1202 http://www.jstor.org/pss/3765550
- ↑ <pubmed> 4132705</pubmed>
- ↑ A S Nadel, J K Green, L B Holmes, F D Frigoletto, B R Benacerraf Absense of need for amniocentesis in patients with elevated levels of maternal serum alpha-fetoprotein and normal ultrasonographic examinations, The New England Journal of Medicine: 1990, 323(9); 557-561 http://www.nejm.org/doi/pdf/10.1056/NEJM199008303230901
- ↑ <pubmed> 6201687</pubmed>
- ↑ <pubmed> 1692998</pubmed>
- ↑ <pubmed> PMC1504442 </pubmed>
- ↑ <pubmed> 7531936</pubmed>
- ↑ <pubmed> 8415426</pubmed>
2010 ANAT2341 Group Projects
Project 1 - Ultrasound | Project 2 - Chorionic villus sampling | Project 3 - Amniocentesis | Group Project 4 - Percutaneous Umbilical Cord Blood Sampling | Project 5 - Fetal Fibronectin | Project 6 - Maternal serum alpha-fetoprotein | Group Assessment Criteria
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 24) Embryology 2010 Group Project 6. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2010_Group_Project_6
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G