2010 Group Project 5
Fetal Fibronectin
Introduction
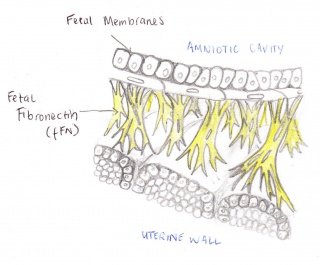
The Fetal Fibronectin (fFN) test measures the amount of secretions of fFN from a woman's vagina and cervix. This technique is used to determine the likelihood of a premature birth occurring. Fetal fibronectin is a protein based plasma that acts as a form of glue attaching the amniotic sac to the uterine wall. Fetal fibronectin is commonly present between 22 to 35 weeks of pregnancy. It is released into the upper vagina towards the onset of labour. If the test finds fFN between this period, the woman will have a chance of going into labour. Hence, if there is no fFN found, it is most likely that they woman will not going into pre-term labour. Therefore, this test allows the woman to see if they are at risk or not.
About pre-term births
History of Fetal Fibronectin
Procedure
Test Results
Targeted woman
The American College of Obstetricians and Gynecologists doesn’t recommend pregnant women who have no symptoms and are not at high risk to be put to test , as the test hasn’t shown the accuracy predicting asymptomic woman.[1]
Only 2 groups of pregnant woman would be recommended to put to test , which are the group that have contraction s or other symptoms of preterm labor in between week 24th and 34th.The other group would be the group that are at high risk for preterm labor, such as women with prior preterm delivery, multifetal gestations, uterine abnormalities and cervical factors , but have no symptoms.[2]
The test should only be used on those pregnant woman who are at high risk and show symptoms of preterm labor to help make a more accurate diagnosis.For example , woman who have intact amniotic membranes, a cervix that has not dilated more than 3 cm, only slight vaginal bleeding, no cervical cerclage(a cervix that has been sewn shut during pregnancy to keep the baby in the uterus).[3]
Reliability
The fFN(fetal fibronectin) remains relatively very low levels throughout the first 22 weeks of gestation.The following dramatically increase of the concentration that reaches 50 ng/mL or over from 22nd week onwards associates with the increase of the incidence of preterm labor before week 37th of gastation.[4] According to a study carried out in a teaching hospital, the presence of fetal fibronectin in the cervicovaginal secretions of preterm birth symptomatic women indicates a significant risk for subsequent preterm birth while the absence of fetal fibronectin in this group of woman is a very strong indication that subsequent preterm birth is unlikely to occur.[5] The findings are ,the preterm birth (before 37 weeks of gestation) rate in the population studied was 19.1%. Fetal fibronectin predicted preterm birth with sensitivity of 63%, specificity of 95.6%, positive predictive value of 77.3%, and negative predictive value of 91.6%. A negative test accurately excluded (97.9%) the chance of subsequent birth during the three weeks interval following sampling.[5]A study with objective to determine the accuracy with which cervico-vaginal fetal fibronectin predicts preterm delivery using systematic quantitative overview of the available literature ,concludes that fetal fibronectin test is the most accurate in predicting spontaneous preterm birth within 7 to 10 days after the test of symptomatic woman before advanced cervical dilatation. A total of 26 876 women involved in the study and among asymptomatic women the best summary likelihood ratio for positive results was 4.01 , 95% confidence interval 2.93 to 5.49 for predicting birth before 34 weeks' gestation, with corresponding summary likelihood ratio for negative results of 0.78. Among symptomatic women the best summary likelihood ratio for positive results was 5.42 for predicting birth within 7-10 days of testing, with corresponding ratio for negative results of 0.25.
Although the test has been commonly used in labour and delivery units to help in the management of preterm labour asymptomatic woman, yet it is not an official screening method to be used of pregnant woman in general, as there is not sufficient evidence to recommend its use as a screening method.Fetal fibronectin test, if combined with clinical findings, has a potentially important role in clinical management of women with symptoms suggestive of preterm labour.[5] Since this review found an association between knowledge of fFN results and a lower incidence of preterm birth before 37 weeks, further research should be encouraged.[6]
Advantages
Infants that born preterm are mainly associated with high chances of mortality and getting health and development problems as they grow up.Complications associated include acute respiratory, gastrointestinal, immunologic, central nervous system problems, hearing, and vision defects, longer-term motor, cognitive, visual, hearing, behavioral, social-emotional, health, and growth problems. The birth of a preterm infant can also bring considerable emotional and economic costs to families and have implications for public-sector services, such as health insurance, educational, and other social support systems.[1] Therefore, being able to predict preterm birth would eliminate those problems listed above.
Disadvantages
There is no side effects in this test, rather the use of fFN test in routine clinical practice allows management and resources to be targeted more appropriately and may limit unnecessary interventions.[7]
Treatment
The current treatment has been focused on inhibiting uterine wall contractions. These interventions do not reduce the incidence of preterm birth but they do delay delivery long enough to allow the administration of antenatal steroids and transfer of the mother and fetus to a hospital where they may receive appropriate care. These interventions have reduced the rates of perinatal mortality and morbidity.[1] According to a study carried out in New Zealand with the objective to compare the treatment received by both fFN positive and fFN negative woman. Women with a positive fFN were more likely to receive antenatal corticosteroids (96.4 vs 4.7%),and tocolysis (71.4 vs 2.4%) and to be admitted antenatally (96.4 vs 54.4%). In short, women with a positive fFN result received different treatment to those with a negative fFN. [7]
Further Research
Glossary
References
- ↑ 1.0 1.1 1.2 Institute of Medicine (US) Committee on Understanding Premature Birth and Assuring Healthy Outcomes; Behrman RE, Butler AS, editors.Washington (DC): National Academies Press (US);2007
- ↑ <pubmed>7738935</pubmed>
- ↑ American College of Obstetricians and Gynecologists (ACOG). Assessment of Risk Factors for Preterm Birth. ACOG Practice Bulletin, number 31, October 2001 (reaffirmed 2008).
- ↑ Akush Ginekol (Sofiia). 2010;49(2):39-42. Bulgarian.
- ↑ 5.0 5.1 5.2 <pubmed>8688390</pubmed>
- ↑ <pubmed>18843732</pubmed>
- ↑ 7.0 7.1 <pubmed>16953860</pubmed>
2010 ANAT2341 Group Projects
Project 1 - Ultrasound | Project 2 - Chorionic villus sampling | Project 3 - Amniocentesis | Group Project 4 - Percutaneous Umbilical Cord Blood Sampling | Project 5 - Fetal Fibronectin | Project 6 - Maternal serum alpha-fetoprotein | Group Assessment Criteria
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 25) Embryology 2010 Group Project 5. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/2010_Group_Project_5
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G