2009 Group Project 4: Difference between revisions
| Line 918: | Line 918: | ||
*Hill M., 2009, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G [http://embryology.med.unsw.edu.au/OtherEmb/mouse1.htm] | *Hill M., 2009, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G [http://embryology.med.unsw.edu.au/OtherEmb/mouse1.htm] | ||
*Bard, J & M Kaufman (2003). 'The Edinburgh Mouse Atlas Project' The Medical Research Council & University of Edinburgh [http://genex.hgu.mrc.ac.uk/Atlas/intro.html] | |||
*The Jackson Laboratory [http://jaxmice.jax.org/news/2009/leukemia.html] | *The Jackson Laboratory [http://jaxmice.jax.org/news/2009/leukemia.html] | ||
Revision as of 17:57, 23 September 2009
THE MOUSE (Mus musculus)
Introduction
The mouse is a small animal which can be effectively used as a model to study embryological development. It belongs to the class mammalian and order rodentia and shares a significant similarity of homology with humans. The mouse is one of the most commonly used animals as model organisms in experimental embryology and most sciences. The mouse is a useful mammalian model for human embryological development because the mouse:
- Has the same size genome as the human genome
- Genes can be easily manipulated and studied
- Have a high degree of homogeny with humans
- Produces a large offspring in a short amount of time
- Are not limited in ethical issues as they can be manipulated in ways which would be unethical to do to humans
- Are small organisms and are easily maintained
- Are not expensive
History of the use of the Mouse Embryo Model
Introduction
The mouse has had a significant contribution to the field of Biomedicine since the middle of the sixteenth century (Hendrich et al. 2004). During the twentieth century, the use of the mouse embryo in particular increased significantly, and continues to be a popular experimental choice for researchers.
It's popularity over time in mammalian biology, biomedicine, immunology, oncology, pathology, and genetics is a result of:
- The small size of the mouse and its resistance to infection (Nagy et.al. 2003).
- The efficient mouse breeding system: Which consistently monitors the characterisitcs, that are precisley known, generation after generation, thereby producing highly standardised strains (Hedrich et al. 2004).
- The metabolic and internal anatomical similarities between the mouse and the human, which allows for comparisons. Because of these similarities, they share similar diseases, for example, cancer, diabetes, autoimmunity, endocrine disease, and neurological dysfunctions (Hedrich et al. 2004).
- The ability to manipulate the mouse germ line. This can be achieved by the:
1. Genetic manipulation of embryonic stem (ES) cells, or
2. Direct injection of cloned DNA into zygotes
Please note that the following findings are based on research which have utilised the model of the mouse embryo.
Gregor Johann Mendel (1822-1884), Augustinian priest and scientist
In his notes, One Hundred Years of Mouse Genetics: An Intellectual History II The Molecular Revolution (1981–2002), Kenneth Paigen wrote that Mendel's first experiment on the transmission of inheritance was made using mice. Due to complaints from members of the Catholic Church about the smell of the mice , he changed his material to Pea plants (Berry 1987).
1895: Robert Heinrich Johannes Sobotta (1869-1945), German Anatomist [1]
Researched the fertilisation and cleavage of the mouse's egg (Sobotta, 1895). Link to Dr. Sobotta's research [2]
What did he find?
- The corpus Luteum is formed by the enlargement of the epithelial cells of the follicle, aided by growth of connective tissue.
- There is no distinction between corpora lutea vera and corpora lutea spuria.
- The corpora lutea do not degenerate. They remain unchanged during the life of the animal, and therefore add to the size of the ovary.
1949: John H. Hammond, Jr. (1888-1965), Animal Husbandry Scientist
What did he do?
Cultured eight-cell morulae and four-cell-stage embryos to the Blastocyst sage. This was the first report of successful attempts to culture mouse embryos in vitro to the Blastocyst stage (Nagy et.al. 2003).
Embryos removed at the two-cell stage soon after died (Nagy et.al. 2003).
1956: Wesley Kingston Whitten (1918-), Australian Veterinary Scientist
What did he do?
Cultured 8-cell mouse embryos to the Blastocyst stage.
How did he do this?
Used a medium containing Krebs-Ringer's bicarbonate solution supplemented with glucose and bovine serum albumin (Nagy et. el. 2003).
Soon after, he found that some two-cell-stage embryos developed into blastocysts, upon some modifications to the original medium.
He moved to the United States later on, continuing his work in the well known, The Jackson Laboratory.
1958: Dame Anne Laura Dorinthea McLaren (1927-2007), Developmental Biologist
Developed the first birth of mice in-vitro. Link to Published article [3]
1962: George Todaro (1937-) and Howard Green, Cell Biologists
- By studying the fibroblast cells of the mouse embryo , they explained how cells behave in a similar fashion once they had undergone transformation (Bhamrah et al. 2002).
How did they do this?
- Todaro and Green cultured the fibroblast cells from the mouse embryo.
What did they find?
- A few weeks after the culture, the growth rate of the Fibroblast cells slowed down. They thought that the cells, as with normal human fibroblast cells, would eventually stop didviding and die. However this was not the case.
- Two to three months later, the cell growth rate increased. This meant that the resulting new population of cells had undergone spontaneous transformation. The resulting permanently dividing cell line, called the '3T3 cell line', have been growing in culture for over 20 years. (Bhamrah et al. 2002)
1963: Ralph L. Brinster (1932-)[4], American Geneticist
What did he do?
- Determined the nutritional requirements of the pre-implantation mouse embryo.
- Established the microdrop technique that allowed two-cell-stage mouse embryos to be cultured to the Blastocyst stage (Nagy et.al. 2003).
1965: Beatrice Mintz (1921-)[5], American Embryologist
What did she do?
- Found that the zona pelucida of the mouse embryo could be digested using pronase (Nagy et.al 2003).
- Generated adult chimeras. Link to description of a chimera [6]
1974: Rudolf Jaenisch (1942-), German biologist and Beatrice Mintz
What did they do?
Injected purified SV40 DNA into the mouse blastocyst cavity.
What was the result?
Viral DNA sequences were detected in the somatic tissue of these mice. This result suggested that the DNA had integrated into the genome of embryonic cells (Nagy et.al. 2003).
1976: Rudolf Jaenisch
What did he find?
Monoloney murine leukemia virus can be introduced into the mouse germ line in a stable manner by viral infection of preimplantation mouse embryos. The significance of this finding is outlined below under the findings of Anderson (1980), Capecchi (1980), and Gordon (1980).
1977: R.J. Mullen
What did he do?
Fused the normal and reeler mouse embryo at the morula stage to produce allophenic mice. (Rouvroit & Goffinet 1998)
1980: Ralph L. Brinster (1932-), American Geneticist
What did he do?
The first to inject purified globin mRNA into mouse zygotes (Nagy et.al. 2003).
This formed the basis for the production of transgenic mice.
1980: Wayne F. Anderson, Lydia Killos, L Sanders-Haigh, P J Kretschmer, and E G Diacumakos & 1980: Mario R. Capecchi
What did they do?
Microinjected cloned herpes simplex virus (HSV) thymidine kinase (tk) gene into the nuclei of cultured TK-deficient mouse fibroblast cells.
Result?
It allowed for the stable incorporation and expression of the tk gene.
Importance?
This suggested that the microinjection of DNA into the one-cell mouse embryo might allow for the efficient introduction of cloned genes into the developing mouse (Nagy et.al. 2003). This triggered researchers, including Gordon mentioned below, to test this possibility.
Click here for Anderson's published article, or click here for Capecchi's published article.
1980: J.W. Gordon, G A Scangos, D J Plotkin, J A Barbosa, and F H Ruddle
What did they do?
Directly injected the cloned gene into the pronuclei of zygotes.
Result?
Introduced the gene into the somatic tissues (Nagy et.al. 2003).
Click here for Gordon's published article.
1981: M.H. Kaufman & M.J. Evans, and 1981: G.R. Martin
What did they do?
They were the first to develop ES cells from cultures mouse blastocysts (Nagy et.al. 2003).
Click here for Martin's published article.
1983: Davor Solter (1939-), Developmental Biologist
What did he do?
Transferred nuclei between zygotes.
The importance?
Revealed the importance of parent gene imprinting in mammalian development (Nagy et.al. 2003).
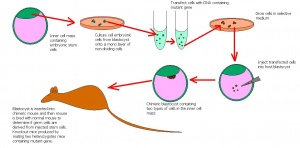
1986: Elizabeth Robertson, Allan Bradley, Michael Kuehn & Martin Evans
What did they do?
They were the first to genetically manipulate ES cells.
Importance?
Allows for the modification and selection of cells with germ-line potential. In turn, this allows them to derive the transgenic strains with pre-determined genetic changes.
By doing this, they inserted many proviral vector sequences that
- Provide new chromosomal molecular markers for linkage studies in the mouse, and
- May cause mutations [7] .
1996: Toshio Ohshima, Jerrold M. Wardt, Chang-Goo Huht, Glenn Longenecker, Veeranna, Harish C.Pant, Roscoe O. Bradyt, Lee J. Martins, and Ashok B. Kulkarni
Generated Cyclin-dependant kinase 5 (Cdk5) null mice (a type of mutant mice) by homologous recombination to assess the role of Cdk5 in vivo. Targeted ES cells were injected into blastocysts to generate chimeric mice.
1998: Teruhiko Wakayama (1967-), Developmental Biologist and Ryuzo Yanagimachi (1928-), Animal Embryologist
What did they do?
Cloned the first mouse, named Cumulina.
1998: Nikola Skreb and colleagues
What did they do?
Showed that the early embryonic ectoderm contains cells capable of contributing to all three germ layers of the mouse fetus.
Use of teratocarcinomas
What are teratocarcinomas?
They are '...gonadal tumors that contain a...mixture of different tissue types, all derived from a population of undifferentiated stem cells known as embryonal carcinoma cells.' (Nagy et.al. 2003)
The use of teratocarcinomas in research complement the studies on pluripotentiality of cells from the normal embryo and allows further study of early embryonic development.
First pioneered by Leroy Stevens (1920-), Mammalian Embryologist and Barry Pierce
Leroy C. Stevens, Jr.:
- The first to identify that male mice of the inbred 129 strain have a low incidence of testicular teratoma arising from primordial germ cells (Link to Stevens and C. C. Little's related article [8] PMID: 16578442).
- The first to identify modifier genes such as ter that increase the frequency of teratomas in the testis and eventually developed an inbred strain (129/Sv).
- Developed the LT strain, in which about 50% of females develop ovarian teratocarcinomas.
Ralph Brinster(1974)[9] PMID: 4610074 , Mintz and Illmensee (1975)[10] PMID: 1059147, and Papaioannou et al. (1975) [11] PMCID: PMC433040 demonstrated that embryonic carcinoma (EC) stem cells could be injected into blastocysts to create adult chimeras that contained normal tissues derived from the EC cells.
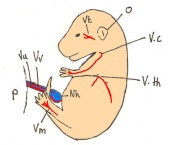
Staging Of Embryonic Development
Reproduction
Mice are polyestrous meaning they can breed many times during the year and can also reproduce all year round. The male and females are capable of breeding at 50 days old and females begin their estrous cycle (go on 'heat') between 25-40 days old. The estrous cycle is 4-5 days in duration and ovulation occurs spontaneously. A mouse produces an average litter of about 10-12 mice and begins a normal estrous cycle 3-10 days following partuition. Having an understanding of mouse reproduction, can highlight the useful nature of studying the mouse as a mamallian model for human emryology.
Theiler stages
Mouse embryonic development commences once the female's egg or oocyte has become fertilized by the male's sperm. Mouse development has a gestation period of 19-21 days, and can range in different strains of mice. The development of an embryo can be categorized into different stages including cell number, somite stages and morphology. The most common method of staging is by Theiler (1989) which categorizes mouse development into prenatal and postnatal stages consisting of 26 and 2 stages respectively. Downs and Davies (1993) have also established a method of staging the mouse development based on morphological changes. Link to Downs and Davies[12]
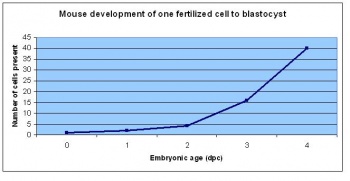
The developmental stages of the mouse embryo can be summarised in the tables and corresponding figures below in relation to characteristics at that stage. The first stage begins with fertiliation of the egg which divides into multiple cells to form a morulla and further go on to form a blastocyst which is ready for implantation.
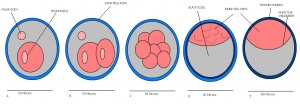
| Theiler Stage | Embryonic Age in Days Post Coitum (dpc) | Stage Characteristic | Cell Characteristics | Zona Pellucida | Location | |
|---|---|---|---|---|---|---|
| 1 | 0-0.9
(range 0-0.25) |
One-celled embryo (fertilized) | One cell | Present | Ampulla | |
| 2 | 1
(range 1-2.5) |
Dividing egg | 2-4 cells
- 1st cleavage after 24hrs |
Present | Travelling down oviduct | |
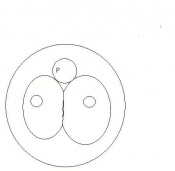
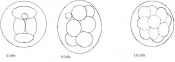
| 3 | 2
(range 1-3.5) |
Morula (early to fully compacted) | 4-16 cells | Present | Oviduct (utero-tubal junction) | |
| 4 | 3
(range 2-4) |
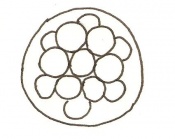
Morula to Blastocyst
-Intra-cellular matrix -Blastocoelic cavity |
16-40 compacted cells
-Inner cell mass -Outer layer of trophectoderm cells |
Present | Uterine lumen | |
| 5 | 4
(range 3-5.5) |
Zona free Blastocyst (hatching) | Blastocyst implants as zona pelludica is lost | Absent | Uterine lumen |
Dr Mark Hill 2009, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
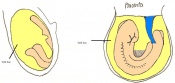
Once the Blastocyst has lost the zona pellucida, it is free to implant onto the uterine wall. After implantation multiple features of a trilaminar embryo start to develop. Ectoderm, endoderm and mesoderm layers form, which further develop many structures corresponding to different stages as summariesed in the table below. The developing mouse embryo which is implanted on the uterine wall can be viewed by ultrasonic radiation. Link to ultrasound of mouse embryo [13]
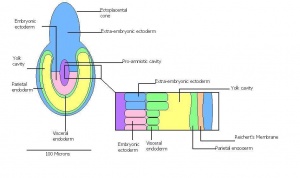
| Theiler Stage | Embryonic age in Days Post Coitum (dpc) | Stage Characteristic | Cell characteristics | |
|---|---|---|---|---|
| 6 | 4.5 (range 4-5.5)
Human carnegie stage: 4 |
Attachment of blastocyst
-Implantation |
Embryonic Endoderm | |
| 7 | 5 (range 4.5-6)
Human carnegie stage: 5 |
Implantation
-Egg cylinder -Ectoplacental cone |
Inner cell mass increases
-Epiblast -Mural trophectoderm lined by endoderm | |
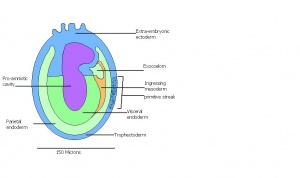
| 8 | 6 (range 5-6.5)
Human carnegie stage: 5 |
embryonic and extra-embryonic regions
-Pro-amniotic cavity |
Trophoblast giant cells invade
-Maternal blood invades -Reichert's membrane | |
| 9 a) | Pre-streak | Advanced Endometrial and egg cylinder
-Embryonic axis |
Embryonic and extra-embryonic ectoderm
-Uterine crypts lose lumen | |
| 9 b) | Early streak | Gastrulation begins | Mesodermal cells | |
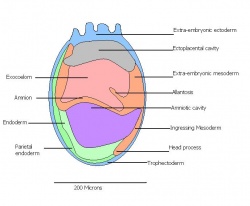
| 10 a) | 7 (range 6.5-7.5)
Mid to late streak Human carnegie stage: 8 |
Amnion formation | Amniotic fold
-Allantoic bud -Primitive node -Amnion closes | |
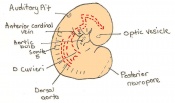
| 11 | 7.5 (range 7.25-8)
Human carnegie stage: 9 |
Neural plate and presomites | Amniotic cavity, exocoelom and ectoplacental cleft
-Allantoic bud elongates -Notochodal plate -Early head fold -Foregut pocket |
This table can be seen in more detail by following the link More on stage 6-11
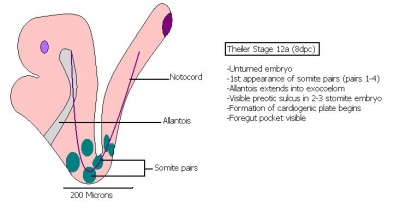
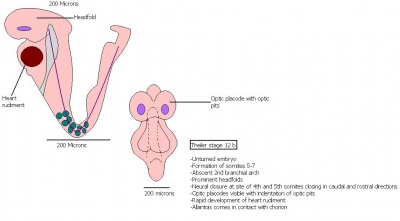
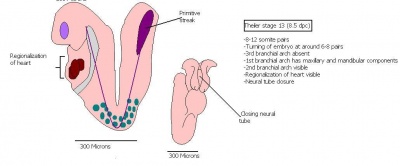
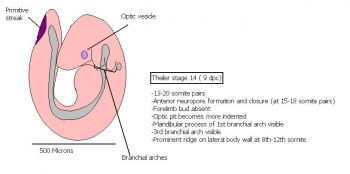
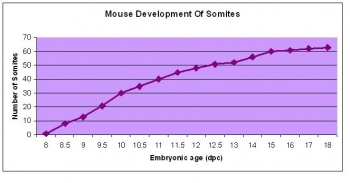
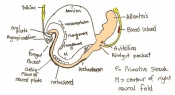
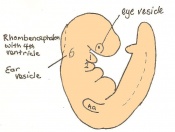
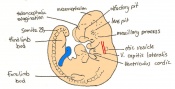
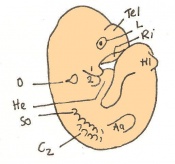
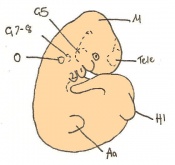
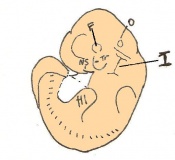
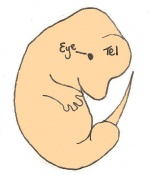
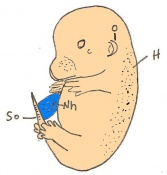
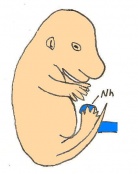
The next stage (Theiler stage 12) is marked by the first somite pair formation. Stages 13 onwards is characterized by increasing somite pairs up to 20 somite pairs. These embryo stages have characteristic features of developing forgut, prominent head fold, optic pits, branchial arch and neuropore formation. These four stages can be summarised in the figures 4-7 below. These stages can be seen in table form by following the link Table of stages 12-14
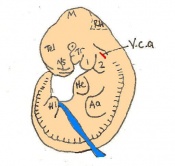
Following stage 14, The embryo continues to develop more distinguished features of a mouse, including forelimb and hindlimb bud, tail elongation, lungs and brain development. The stages 15-20 are summarised in the table below.
| Theiler Stage | Embryonic Age (dpc) | Stage Characteristic | Cell Characteristic | Somite Number(pairs) | |
|---|---|---|---|---|---|
| 15 | 9.5 (range 9-10.25)
Human carnegie stage: 12 |
Forelimb bud (8-12th somite pair)
-Posterior neuropore |
Hind limb bud
-Telencephalic and diencephalic vesicle division -Lung development -Dorsal pancreatic bud |
21-29 | |
| 16 | 10 (range 9.5-10.75)
Human carnegie stage: 13-15 |
Caudal neuropore closes
-Hind limb and tail bud |
Concave 3rd and 4th branchial arches.
-Rathke's pouch -Nasal processes -Ventral pancreatic bud |
30-34 | |
| 17 | 10.5 (range 10-11.25)
Human carnegie stage: 13-15 |
Deep Lens Indentation | Physiological umbilical hernia
-1st branchial arch (maxillary and mandibular components) -Brain tube development -Tail elongates |
35-39 | |
| 18 | 11 (range 10.5-11.25)
Human carnegie stage: 13-15 |
Lens Vesicle closes | Cervical somites not visible
-Brain rapidly grows -Nasal pit |
40-44 | |
| 19 | 11.5 (range 11-12.25)
Human carnegie stage: 16 |
Lens vesicle separated
–Closed and detached from ectoderm |
Eyes and their peripheral margins
-Limb-girdle and arm -Anterior footplate. -Auditory hillocks |
45-47 | |
| 20 | 12 (range 11.5-13)
Human carnegie stage: 17 |
Gingers | Anterior footplate (develops angles)
-Posterior footplate -Pigmentation of retina -Tongue and brain vesicles |
48-51 |
This table can be seen in more detail by following the link More on stage 15-20 To visualize the mouse development so far a movie showing the mouse embryo can be seen by following the link [14]( note the heart beat)
The last few stages of develop (stages 21-26) shown in table 4 are the complete prenatal developmental stages. These stages involve fine development of the pinna over the external acoustic meatus, finger and toe,hair, eyes and whiskers.
| Theiler Stage | Embryonic Age (dpc) | Stage Characteristic | Cell Characteristic | Somite Number(pairs) | |
|---|---|---|---|---|---|
| 21 | 13 (range 12.5-14)
Human Carnegie Stage 18-19 |
Anterior footplate indented
-Pinna develops |
Digits, elbow and wrist
-5 rows of vibrissae visible -Hair follicle over eye and ear -Lens loses lumen |
52-55 | |
| 22 | 14 (range 13.5-15)
Human Carnegie stage 20-23 |
Individual fingers on anterior footplate
-Umbilical hernia visible |
Deep indentations between toes( not separated)
-Long bones of limbs present -Hair present in pectoral, trunk and pelvis -Pinna turned forwards |
56-60 | |
| 23 | Human fetal period | Toes separate | Hair follicles present in cephalic region
-Eyelids open -Pinna covering ½ of external auditory meatus |
>60 | |
| 24 | 16 | Reposition of umbilical hernia | Parallel fingers (2-5)
-Toe nail primordia -Eyelids fused -Complete coverage by pinna -Increase in peritoneal sac size |
>60 | |
| 25 | 17 | Skin thickened and wrinkled
-Umbilical hernia disappeared |
Subcutaneous veins less visible
-Fingers and toes parallel -Whiskers visible |
||
| 26 | 18 | Skin thickened
Long whiskers |
Pinna larger (auditory meatus lumen not visible) |
To view the mouse embryo in late stages of development clink the link [[15]]
Following the prenatal development are the two Theiler stages 27 and 28 which involve birth of the mouse and postnatal development.
Timeline of Development
The development of the mouse embryo takes 19 days on average from fertilisation to newborn mouse. This timing depends on rate of development.
- Development of the Mouse Embryo
Genetics
Genome
Sequencing of the mouse genome was completed in late 2002. Tha haploid genome is about 3 billion long (3000 Mb distributed over 20 chromosomes) and therefore equal to the size of the human genome. The current estimated gene count is 23,786 and humans are estimated to have 23,686 genes.
The genetic map of the mouse
Genetic maps, the road maps of genetics, are of two types: linkage and physical. The 'sign posts' on the maps are loci, any location or marker in the genome that can be detected by genetic or DNA analysis. The term 'gene' is more restrictive than loci and refers to DNA segments that encode proteins or can be linked to phenotypes. Linkage maps are recombinational maps and are constructed by carrying out linkage crosses that measure the recombination frequency between genes or loci on the same chromosome.
- The first genetic linkage in the mouse (and first autosomal linkage in mammals) was described in 1915 in the classic paper on the linkage of pink-eyed dilution and albino (Haldane et al., 1915)
- Genetic mapping with spontaneous mutations that created visible phenotypes, such as changes in coat colour/texture or behaviour was labarious and sometimes took years because crosses between mice carrying recessive mutations yielded so few informative progeny, and genes on only one or two chromosomes could be scored in each cross.
- The first real breakthrough in linkage mapping, enabling the scoring of many test markers and chromosomes in the same cross, was the discovery and use of co-dominant biochemical (isoenzyme) genes (e.g. glucose phosphate isomerase 1, Gpil; Hutton and Coleman 1969).
How large is the genome?
With the use of Feulgen reagent the quantitative DNA-specific staining can be achieved. Through micro photometric measurements of the staining intensity in individual sperm nuclei, it is possible to determine the total amount of DNA present in the haploid mouse genome (Laird, 1971). Measurements indicated a total haploid genome content of 3 pg, which translates into a molecular weight of 1.8 x 1012 daltons (Da).
How complex is the genome?
Another method for determining genome size relies upon the kinetics of DNA renaturation as a sign of the total content of different DNA sequences in a sample. When a solution of double stranded DNA is denatured into single strands which are then allowed to renature, the time required for renaturation is directly proportional to the complexity of the DNA in the solution, if all other parameters are held constant.
Complexity is a measure of the information contained within the DNA. The maximal information possible in a solution of genomic DNA purified from one animal or tissue culture line is equivalent to the total number of base pairs present in the haploid genome.The information content of a DNA solution is independent of the actual amount or concentration of DNA present. DNA obtained from one million cells of a single animal or cell line contains no more information than the DNA present in one cell. Furthermore, if sequences within the haploid genome are duplicates of one another — repeated sequences — the complexity will drop accordingly.
Renaturation analysis of mouse DNA reveals an overall complexity of approximately 1.3-1.8 x 109 bp. This value is only 40-60% of the size of the complete haploid genome and it implies the existence of a large fraction of repeated sequences.
What proportion of the genome is functional?
Bacterial species are remarkably efficient at packing the most genetic information into the smallest possible space. In one analysis of a completely sequenced 100 kb region of the E. coli chromosome, it was found that 84% of the total DNA content was actually used to encode polypeptides. Most of the remaining DNA is used for regulatory purposes, and only 2% was found to have no recognizable function.
Comparative sequence analysis over long regions of the mouse and human genomes shows evolutionary conservation over stretches of sequence that do not have coding potential or any obvious function (Hood, 1992). However, sequences can only be conserved when selective forces act to maintain their integrity for the benefit of the organism. Thus, conservation implies functionality.The fraction of the mouse genome that is functional is likely to lie somewhere between 5% and 10% of the total DNA present.
Chromosome number and banding patterns
All of the Mus musculus subspecies have the same standard karyotpye with;
- 20 pairs of chromosomes, including 19 autosomal pairs and the X and Y chromosomes
- All of the 19 autosomes as well as the X and Y chromosome appear to be telocentric, with a centromere at one end and a telomere at the other.
The biological explanation for this uniformity in chromosome morphology is entirely unknown; however, it makes the task of individual chromosome identification much more difficult than it is with human karyotypes.
Chromosome length and DNA content
The amount of DNA present in each chromosome can be estimated by measuring its length — cytologically — relative to the sum of the lengths of all 20 chromosomes and multiplying this fraction by the total genome length of 3,000 mb (Evans, 1989). From these measurements, one finds that the largest chromosome (1) has a DNA length of approximately 216 mb and the smallest chromosome (19) has a DNA length of 81 mb, with all others following in a near-continuum between these two values.
Mice with chromosomal aberrations
The diploid chromosomal complement of standard inbred laboratory strains is 2N=40:19 autosomes, X and Y chromosomes.The autosomes and the X chromosomes are telocentric (i.e. the centromere is at one end of a single-armed chromosome) while the y chromosome is acrocentric (i.e. it has a short p arm as well as the longer q arm, use of 'p' and 'q' is patterned on human chromosomal nomenclature). The sex determining genes resides in the short arm of the Y chromosome.
Strains of mice whose chromosomal complement devaites from the normal chromosomal makeup are designated chromosomal aberration starins. Chromosomal aberrations can include intra- and interchromosomal rearragements or aneuploidy. These include;
- inversions and transpositions, rearrangements of DNA segments within chromosomes
- reciprocal translocations, Robertsonian chromosomes, and insertions, exchanges of DNA segments between chromosomes
- aneuploidy, deviations from the normal diploid number of chromosomal arms in somatic cells (e.g. trisomies).
Some chromosomal deletions and duplications also may be cytologically detectable.
Comparative mapping
Comparative mapping began in the early 1970s and gained momentum until it culminated with the sequencing of the two genomes in 2001 and 2002.
- First conserved mouse and human autosomal linkage was reported in 1978.
- 13 conserved autosomal segments and estimated 178(+-)39 chromosomal rearrangements between mouse and human chromosomes were identified (Nadeau and Taylor,1984)
- Sequencing of the two genomes revealed that 95% of the coding sequence is conserved at the DNA level (Consortium,2002)
The Knockout Mouse
A knockout mouse is a genetically engineered mouse in which one or more genes have been turned off through a gene knockout. They are important animal models for studying the role of genes which have been sequenced. Mice are currently the most closely related laboratory animal species to humans for which the knockout technique can easily be applied.The first knockout mouse was created by Mario R, Capecchi, Martin Evans and Oliver Smilthies in 1989. [[16]]
- Use
Knocking out the acitivity of a gene provides information about what that gene normally does. Humans share many genes with mice. Consequently, observing the characterisitcs of knockout mice gives researchers information that can be used to better understand how a similar gene may contribute to disease in humans.
- Areas of research in which knockout mice have been useful include:
- cancer
- obesity
- heart disease
- diabetes
- arthritis
- anxiety
- aging
- Parkinson's disease.
- Limitations of the use of Knockout mice
While knockout mice technology represents a valuable research tool, its use may be limited. For example:
- About 15% of gene knockouts are developmentally lethal, which means that the genetically altered embryos cannot grow into adult mice. This problem is often overcome through the use of conditional mutations. The lack of adult mice limits studies to embryonic development and often makes it more difficult to determine a gene's function in relation to human health.
- Knocking out a gene may fail to produce an observable change in a mouse or may even produce different characteristics from those observed in humans in which the same gene is inactivated. E.g mutations in the p53 gene are associated with more than half of human cancers and often lead to tumors in a particular set of tissues. However, when the p53 gene is knocked out in mice, the animals develop tumors in a different array of tissues.
Examples of transgenic models for human diseases
[[17]]
Models for AIDS study
The HIV-1 virus is known to have two major receptors in human cells CD4 and CCR5. Transgenic rabbits expressing the human CD4 gene were used, the virus replicated in rabbit cells but was unable to generate any disease. Therefore, mouse models were generated. It appeared that transgenic rats expressing all the HIV-1 genes except gag and pol showed pathogeny having many similarities to human AIDS.
Models for aging
Aging is a complex phenomenon which has only been partially described. Defects in genomes appear to be a major cause of aging. A growing number of trangenic models are being used to study aging. Mice in which the XPD gene has been knocked out are more sensitive to oxidative DNA damage. This sensitivity was increased further when the XPA gene was also knonked out. These models reflect some of the aging syndromes in human.
Models for Cancer
Transgenic mice are used to generate models for cancer study. The first oncomouse expressed c-myc gene in the mammary gland. This was sufficient to trigger the formation of mammary tumors. Further studies made it pissible to identify additional genes involve din mammary cancer. Genes whose expression is amplified in mammary tumors have an oncogenic effect when used as transgenes. Crossing mice harboring different oncogenes and having knocked out genes has made it possible to determine the cooperative actions of some of these genes.
The Future
The future of the mouse in genome analysis and as a model organism seems virtually unlimited. Whole genome sequence and gene preditction programs make it quite feasible to knock out or genetically modify every mouse gene. Almost certainly, ES cells will play a key role in future mouse genomics because they can be manipulated in culture. More phenotype screens that can detect mutations prior to making live mice will be needed to increase the number and types of mutations that can be detected in the ES cells themselves. While other model organisms, such as Drosophilia, yeast, worms and zebrafish may be easier to manipulate and allow analysis that require hundreds or thousands of animals, the mouse is likely to continue to be the premier mammalian model for understanding human inherited diseases.
International Mouse Strain Resource (IMSR)[18]
An on-line database that lists the international availability and supply of inbred, mutant, and genetically engineered mice.
Current Research
Overview
Mice are highly used models of developmental biology, immunity, neurobiology, and human diseases in pathology. It's main contribution to developmental biology has been through transgenic and knockout technology which are both very important to the research of early development or organogenesis (Slack, 2006).
Examples of the use of Knockout Mouse
1) National Institute of Dental and Craniofacial Research (NIDCR)
2) The International Knockout Mouse Consortium (IKMC)
There goal is to mutate all protein-coding mouse genes using a combination of gene trapping and gene targeting in mouse embryonic stem (ES) cells.
Programs running under IKMC:
- Knockout Mouse Project (KOMP) (USA)
- European Conditional Mouse Mutagenesis Program (EUCOMM) (Europe)
- North American Conditional Mouse Mutagenesis Project (NorCOMM) (Canada)
- Texas A&M Institute for Genomic Medicine (TIGM) (USA)
3) Department of Experimental Medicine, Histology and Embryology Unit, University of Pavia, Pavia, Italy.
In 2009, Al Cornaglia, A Casasco, M Casasco, F Riva and V Necchi used the knock out mouse to research how mutations in the diastrophic dysplasia sulphate transporter (dtdst) gene cuased different forms of the disease chondrodysplasia in humans. By producing a knouck out mice strain of this mutated dtdst gene, they were able to investigate how this gene affected tissue organisation, matrix structure and cell differentiation in the epiphyseal growth plate of the bone. Click here for their published article.
4) In 2003, Dr. Ashok Kulkarni and his colleagues created a mouse model of the disease known in humans as dentinogenesis imperfecta III. This heredtitary tooth disorder involves the teeth wearing down to the innermost pulp.
- They achieved this by knocking out the dentin sialophosphoprotein (Dspp) mouse gene, thought to be responsible for coordinating the mineralization of a tooth's dentin.
- Results: The role of Dspp is to organise the in organising the events during dentin mineralization, including potential regulation of proteoglycan levels. PMID for Dr. Ashok Kulkarni published article: 12721295
A Pubmed search of the use of the mouse as a model in the study of embryology was done which produced 5213 literature articles. To view this search please click here . A brief selection of these article include:
- 'Maternal diabetes alters transcriptional programs in the developing embryo' by Gabriela Pavlinkova, J Michael Salbaum and Claudia Kappen in 2009. They used the mouse as a model to investigate how maternal diabetes alters transcriptional programs in the developing embryo. Maternal diabetes is a known risk factor for abnormalities in the newborn, but it is unknown how this comes about. They hypothesised that the defects were caused by a change in critical developmental pathways that can result in changes in gene expression. Through using a mouse diabetes model, they found that they could provide support for their hypothesis through their results which concluded that exposure to maternal diabetes had caused alterations in the transcriptional profiles of the developing embryo.
- 'Fibroblast Growth Factor 18 Gives Growth and Directional Cues to Airway Cartilage' by Ravindhra G. Elluru, Felisa Thompson and Alisa Reece from the Division of Pediatric Otolaryngology - Head and Neck Surgery at the Cincinnati Children's Hospital in Cincinnati, Ohio, in 2009. These scientists hypothesised that the rate and direction of chondrocyte growth was affected by Fibroblast growth factor 18 (FGF18). They used the mouse as a model and observed the affect that FGF18 had on the cartilage specifying gene, Sox9. They concluded that FGF18 did indeed regulate the rate and direction of chondrocyte growth by up- regulating Sox9 expression.
- 'Epithelial-Mesenchymal Crosstalk in Wolffian Duct and Fetal Testis Cord Development' by Denise R. Archambeault, Jessica Tomaszewski, Avenel Joseph, Barry T. Hinton and Humphrey Hung-Chang Yao from the Department of Veterinary Biosciences, College of Veterinary Medicine, University of Illinois at Urbana-Champaign, Illinois and the Department of Cell Biology, School of Medicine, University of Virginia, Charlottesville, Virginia in 2009. They used the mouse as a model organism to study the epithelial - mesenchymal crosstalk that occurs in Wolffian duct and testis cord development. They found that the epithelial - mesenchymal interactions in the development of these two male structures are critical for normal development. They were able to identify Inhba and its protein product activin A as being an interstitially derived factor that acts upon sertoli cells in the testis that hints that crosstalk occurs between both organs.
Examples of current research
- Fardin et. al. (2009) has used the mouse as a model for understanding the function of oncogenes involved in epithelial-mesenchymal uncontrolled growth. The study examined a protein Dbl which belongs to the family of proto-oncogenes or proteins involved in normal cell growth. These proteins when mutated become oncogenes and cause unregulated cell growth. The transgenic mice where used to incorporate the complement DNA strand of Dbl oncogene for analysis. The study found that expression of onco-Dbl in mice correlated with up regulation of epithelial transition, apoptosis, vasculogenesis and cell proliferation.
- Andreasen et. al. (2009) has used the mouse as a model of immune response to respiratory tract infections by the bacteria Bordetella pertussis. When infected with the bacteria, it secretes a virulence factor called pertussis toxin. This toxin is responsible for promoting bacterial growth and proliferation in the airways. The study showed that the toxin rather than the bacterial cell numbers is important in the production of cytokine and chemokine responses, and hence the pertussis toxin produces the host immune response not the bacteria.
- Regales et. al. (2009) used transgenic mice as models for testing anticancer drugs in lung tumours involved with tyrosine receptor mutations. The lung tumours mutations were of epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor resistance mutations T790M. The mice bearing the EGFR mutation were used to evaluate the effectiveness of a variety of anticancer drugs. Two anticancer agents BIBW-2992 tyrosine kinase inhibitor and EGFR specific antibody cetuximab were tested on the mice containing EGFR mutated lung tumors. It was found that the combination of the two agents produced marked shrinkage of tumours with T790M mutations.
- Gram et. al. (2009) has used mice in development of HIV-1 vaccines. A vaccine needs to have several factors including several antigens and epitopes as well as an appropriate adjuvant capable for inducing a strong host cellular immune response. Transgenic mice have been used to develop a new lipophillic adjuvant called CAF01 which has been studied to assist in generating antibody and CD4 T helper cell responses. This process can generate CD8 T lymphocytes against HIV-1 derived cytotoxic T lymphocyte epitope peptides in human leukocyte antigen mouse models.
Green Lab, School of Medicine, Harvard University[19]
Researchers from the Green Lab:
- cultivated Fibroblastic cells of a 12-13 day mouse embryo by 27 consecutive transfers and roughly 63 cell generations.
- This resulted in a new 'immortalised' cell line called, MMM which:
1.Supports the multiplication of the H9 cells better than the 3T3 line.
2.More effectively maintains the 'immortalised' cells as stem cells.
- Published Article: An immortalized drug-resistant cell line established from 12-13 day mouse embryos for the propagation of human embryonic stem cells. Link to Abstract [20] or [21]
- Article Date: June 28, 2006.
The Jackson Laboratory[22]
How did they do this?
Conducted a series of transplantation experiments using three JAX® Mice strains: C57BL/6J (B6, 000664), B6.129S2-Alox5tm1Fun/J (004155) - an Alox5-deficient mouse on a B6 background, and B6.SJL-Ptprca Pepcb/BoyJ (002014) (also known as "pep boy").
What did they find?
Alox5 deficiency blocks differentiation, alters the cell cycle, and induces apoptosis of long term LSCs (LT-LSCs). At the moment, it is not known why the Alox5 signaling pathway does not have the same effects on mormal HSCs.
Importance?
Their results demonstrate that targeting a specific gene can completely inhibit only cancer stem cells in vivo.
What's next?
The efficiency of an anti-stem cell strategy as a cancer therapy will be clinically trialed in order to target ALOX5 in human leukemia patients (The Jackson Laboratory 2009).
References
- Andreasen C, Powell DA, Carbonetti NH (2009) Pertussis Toxin Stimulates IL-17 Production in Response to Bordetella pertussis Infection in Mice. PLoS ONE 4(9): e7079. doi:10.1371/journal.pone.0007079
- Bard, Kaufman, Dubreuil, Brune, Burger, Baldock, Davidson (1998). An internet accessible database of mouse development anatomy based on a systemic nomenclature, Mechanisms of development, (74) 111-120.
- Bhamrah H.S., & Juneja K. (2002). Molecular Cell Biology. 1st ed. New Delhi: Anmol Publications.
- Fardin P., Ognibene M., Vanni C., et al. (2009) Induction of Epithelial Mesenchimal Transition and Vasculogenesis in the Lenses
of Dbl Oncogene Transgenic Mice. PLoS ONE 4(9): e7058. doi:10.1371/journal.pone.0007058
- Gram G.J., Karlsson I., Agger E.M., Andersen P., Fomsgaard A. (2009) A Novel Liposome-Based Adjuvant CAF01 for Induction of CD8+ Cytotoxic TLymphocytes(CTL) to HIV-1 Minimal CTL Peptides in HLA-A*0201 Transgenic Mice. PLoS ONE 4(9): e6950. doi:10.1371/journal.pone.0006950
- Hedrich H., Bullock G., & Petrusz P. (2004). The Laboratory Mouse. London: Elsevier Academic Press. Click herefor the online text.
- Hill M., 2009, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G [23]
- Bard, J & M Kaufman (2003). 'The Edinburgh Mouse Atlas Project' The Medical Research Council & University of Edinburgh [24]
- The Jackson Laboratory [25]
- Regales et. al. (2009). Dual targeting of EGFR can overcome a major drug resistance mutation in mouse models of EGFR mutation in lung cancer. journal of clinical investigation. doi:10.1172/SC138746
- Rouvroit C.L., & Goffinet A.M. (1998). Advances in Anatomy, Embryology, and Cell Biology: The Reeler Mouse as a Model of Brain Development. Berlin: Springer.
- Nagy A., Gertsenstein M., Vintersten K., & Behringer R. ( 2003). Manipulating the Mouse Embryo: A Laboratory Manual. 3rd ed. New York: Cold Spring Harbor Laboratory Press. Click herefor the online text.
- Theiler K, (1989). ‘The House Mouse; Atlas of Embryonic Development’, Springer - Verlag New York Inc, New York
- Cornaglia Al et. al. 'Dysplastic histogenesis of cartilage growthby alteration of sulphation pathway: a transgenic model'. Connective Tissue Research, 2009;50(4):232-42 PMID: 19637059
- Pavlinkova, G, J M Salbaum & C Kappen. 'Maternal diabetes alters transcriptional programs in the developing embryo'. BMC Genomics. 2009;10:274 PMCID: PMC2715936
- Elluru, R G., F Thompson & A Reece. 'Fibroblast Growth Factor 18 Gives Growth and Directional Cues to Airway Cartilage' Laryngoscope. 2009; 199(6):1154 - 65. PMID: 19358209
- Arcambeault D R., J Tomaszewski, A Joseph, B T. Hinton & H HC Yao. 'Epithelial-mesenchymal crosstalk in Wolffian duct and fetal testis cord development'. Genesis; 47(1): 40-48 PMID: 18979542
- Mouse Genetics, concepts and applications http://www.informatics.jax.org/silver/
ANAT2341 group projects
Project 1 - Rabbit | Project 2 - Fly | Project 3 - Zebrafish | Group Project 4 - Mouse | Project 5 - Frog | Students Page | Animal Development