Talk:Lecture - Week 3 Development
2018
Mark Hill (talk) 10:44, 30 July 2018 (AEST) Reformatted referencing.
J Cell Physiol. 2018 Sep;233(9):7120-7133. doi: 10.1002/jcp.26528. Epub 2018 Mar 25.
Gao LR1,2, Wang G1,2, Zhang J1, Li S1, Chuai M3, Bao Y4, Hocher B1,5, Yang X1,2. Author information Abstract An association has been proved between high salt consumption and cardiovascular mortality. In vertebrates, the heart is the first functional organ to be formed. However, it is not clear whether high-salt exposure has an adverse impact on cardiogenesis. Here we report high-salt exposure inhibited basement membrane breakdown by affecting RhoA, thus disturbing the expression of Slug/E-cadherin/N-cadherin/Laminin and interfering with mesoderm formation during the epithelial-mesenchymal transition(EMT). Furthermore, the DiI+ cell migration trajectory in vivo and scratch wound assays in vitro indicated that high-salt exposure restricted cell migration of cardiac progenitors, which was caused by the weaker cytoskeleton structure and unaltered corresponding adhesion junctions at HH7. Besides, down-regulation of GATA4/5/6, Nkx2.5, TBX5, and Mef2c and up-regulation of Wnt3a/β-catenin caused aberrant cardiomyocyte differentiation at HH7 and HH10. High-salt exposure also inhibited cell proliferation and promoted apoptosis. Most importantly, our study revealed that excessive reactive oxygen species(ROS)generated by high salt disturbed the expression of cardiac-related genes, detrimentally affecting the above process including EMT, cell migration, differentiation, cell proliferation and apoptosis, which is the major cause of malformation of heart tubes. KEYWORDS: cardiac progenitor migration and differentiation; chick embryo; heart tube; high salt; reactive oxygen species PMID: 29574800 DOI: 10.1002/jcp.26528
Curr Opin Cell Biol. 2017 Oct;48:33-39. doi: 10.1016/j.ceb.2017.04.006. Epub 2017 Jun 3. Regulation of gastrulation movements by emergent cell and tissue interactions.PMID: 28586710 Williams ML1, Solnica-Krezel L2. Author information Abstract It is during gastrulation that the primordial germ layers are specified, embryonic axes become morphologically manifest, and the embryonic body plan begins to take shape. As morphogenetic movements push and pull nascent tissues into position within the gastrula, new interactions are established between neighboring cells and tissues. These interactions represent an emergent property within gastrulating embryos, and serve to regulate and promote ensuing morphogenesis that establishes the next set of cell/tissue contacts, and so on. Several recent studies demonstrate the critical roles of such interactions during gastrulation, including those between germ layers, along embryonic axes, and at tissue boundaries. Emergent tissue interactions result from - and result in - morphogen signaling, cell contacts, and mechanical forces within the gastrula. Together, these comprise a dynamic and complex regulatory cascade that drives gastrulation morphogenesis. PMID: 28586710 PMCID: PMC5835147 [Available on 2018-10-01] DOI: 10.1016/j.ceb.2017.04.006
2017
2016
2015
Link added after Lecture.
Links only work with currently enrolled UNSW students.
| ECHO360 Recording |
|---|
|
Lecture 4 - Rich Media Playback | Vodcast Playback | Podcast Playback Links only work with currently enrolled UNSW students. |
- File:ANAT2341 Lecture 3 - Beverdam - Week 3.pdf 2013 Lecture Lecture Date: 2012-08-13 Lecture Time: 16:00 Venue: Biomedical Theatre E Speaker: Annemiek Beverdam
| 2011 Lecture Link | MP3 | Quicktime |
| Week 3 Development | Part 1 | Part 2 | Part 1 | Part 2 |
| Lectopia Lecture Audio |
Introduction
This lecture will continue from the second week into the third week and discuss early placentation, gastrulation and notochord formation. Note that we will be covering only the early events of placentation and a later lecture will cover this topic in more detail.
| File:Week2_001_icon.jpg</wikiflv> | This animation shows the process of implantation, occurring during week 2 of development in humans. |
Lecture Overview
- Understand broadly the events of week 2-3 of human development
- Understand the process early placentation, villi formation
- Understand the process of gastrulation
- Understand the process of axis formation
Early Placentation
Development Animation - Implantation
The trophoblast layer has now differentiated into two morphologically distinct cellular layers.
- Syncitiotrophoblasts - form a multinucleated cytoplasmic mass by cytotrophoblast cell fusion and both invade the decidua and secrete hCG
- Cytotrophoblasts - form a cellular layer around the blastocyst, proliferates and extends behind syncitiotrophoblasts
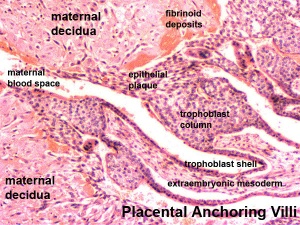
Early Utero-Placental exchange - transfer of nutrition from maternal lacunae filled with secretions from uterine glands and maternal blood from blood vessels. The development of trophoblast villi extending into the uterine decidua.
There are three stages of villi development:
- Primary Villi - cytotrophoblast
- Secondary Villi - cytotrophoblast + extraembryonic mesoderm
- Tertiary Villi - cytotrophoblast + extraembryonic mesoderm+ blood vessels
There are two main types of early villi:
- Anchoring villi - attached to decidua
- Floating villi - not attached to decidua, floating in maternal lacunae.
Maternal endometrium
- Decidual reaction
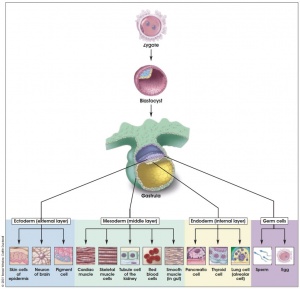
Gastrulation
Development Animation - Mesoderm | Gastrulation
Gastrulation, (Greek = belly) means the formation of gut, but has been used in a more looser sense to to describe the formation of the trilaminar embryo. The epiblast layer, consisting of totipotential cells, derives all 3 embryo layers: endoderm, mesoderm and ectoderm. The primitive streak is the visible feature which represents the site of cell migration to form the additional layers.
Historically, gastrulation was one of the earliest observable morphological event occurring in the frog embryo. Currently, the molecular and physical mechanisms that regulate patterning and migration during this key event are being investigated in several different animal models. In humans, it is proposed that similar mechanisms regulate gastrulation to those found in other vertebrates.
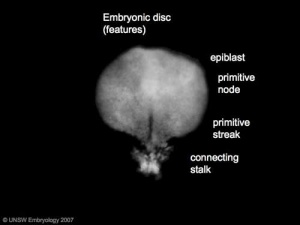
- primitive node - region in the middle of the early embryonic disc epiblast from which the primitive streak extends caudally (tail)
- nodal cilia establish the embryo left/right axis
- axial process extends from the nodal epiblast
- primitive streak - region of cell migration from the epiblast layer forming sequentially the two germ cell layers (endoderm and mesoderm)
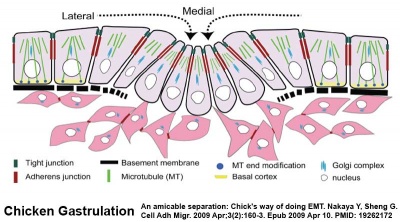
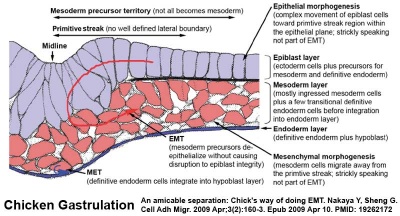
Epithelial to Mesenchymal Transition
|
|
Epithelial cells (organised cellular layer) which loose their organisation and migrate/proliferate as a mesenchymal cells (disorganised cellular layers) are said to have undergone an Epithelial Mesenchymal Transition (EMT). Mesenchymal cells have an embryonic connective tissue-like cellular arrangement, that have undergone this process may at a later time and under specific signaling conditions undergo the opposite process, mesenchyme to epithelia. In development, this process can be repeated several times during tissue differentiation.
This process occurs at the primitive streak where epiblast cells undergo an epithelial to mesenchymal transition in order to delaminate and migrate.
MH - there are a number of common cellular changes that occur during embryonic development at different times and in different tissues, which we can classify into "developmental mechanisms".
Notochord
Development Animation - Notochord
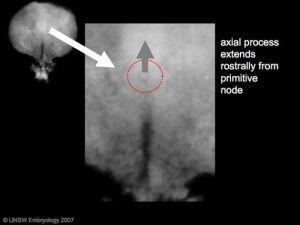
The notochord is a structure which has an early mechanical role in embryonic disc folding and a major signaling role in patterning surrounding embryonic tissue development. This signaling role patterns many different tissues (neural plate, neural tube, somites, endodermal organs). It has its own sequence of development from a primitive axial process and is a developmental feature not present in the adult anatomy.
- axial process an initial epiblast hollow epithelial tube which extends in the midline from the primitive pit, cranially in the embryonic disc (toward the oral membrane).
- neuroenteric canal is a transient communication between the amnionic cavity and the yolk sac cavity formed by the axial process.
- notochordal plate forms from the axial process merging with the endoderm layer.
- notochord forms from the notochordal plate which then separates back into the mesoderm layer as a solid column of cells lying in the midline of the embryonic disc and running rostro-caudally (head to tail).
- An alternate name for the notochord is "axial mesoderm".
MH - Much of our knowledge of this structure comes from the study of animal models of development.
References
Textbooks
- The Developing Human: Clinically Oriented Embryology (8th Edition) by Keith L. Moore and T.V.N Persaud - Chapter 3
- Larsen’s Human Embryology by GC. Schoenwolf, SB. Bleyl, PR. Brauer and PH. Francis-West - Chapter 3
Online Textbooks
- Developmental Biology by Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000 Figure 11.22. The cleavage of a single mouse embryo in vitro | Figure 11.25. Mouse blastocyst hatching from the zona pellucida | Figure 11.20. Development of a human embryo from fertilization to implantation | Figure 11.24. Implantation of the mammalian blastocyst into the uterus
- Molecular Biology of the Cell 4th ed. Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter New York and London: Garland Science; c2002 - Fertilization | Figure 21-84. Scanning electron micrographs of the early mouse embryo | Figure 21-69. The blastula
- Molecular Cell Biology by Lodish, Harvey; Berk, Arnold; Zipursky, S. Lawrence; Matsudaira, Paul; Baltimore, David; Darnell, James E. New York: W. H. Freeman & Co.; c1999 Chapter 13. Regulation of the Eukaryotic Cell Cycle
- The Cell - A Molecular Approach by Cooper, Geoffrey M. Sunderland (MA): Sinauer Associates, Inc.; c2000 Figure 14.37. Meiosis of vertebrate oocytes
- HSTAT - In Vitro Fertilization As A Medical Treatment For Male or Female Infertility
- Human Molecular Genetics 2 Strachan, Tom and Read, Andrew P. New York and London: Garland Science; c1999 Figure 8.19. Changes in DNA methylation during mammalian development
Search
- Bookshelf placentation | placenta | implantation | gastrulation | notochord
- Pubmed placentation | placenta | implantation | gastrulation | notochord
Archive Links
- Week 3 Slides 2009 | Lecture 4 2008 | Lecture 4 2008 slides