Talk:ASA Meeting 2013 - Placenta
Additional Information
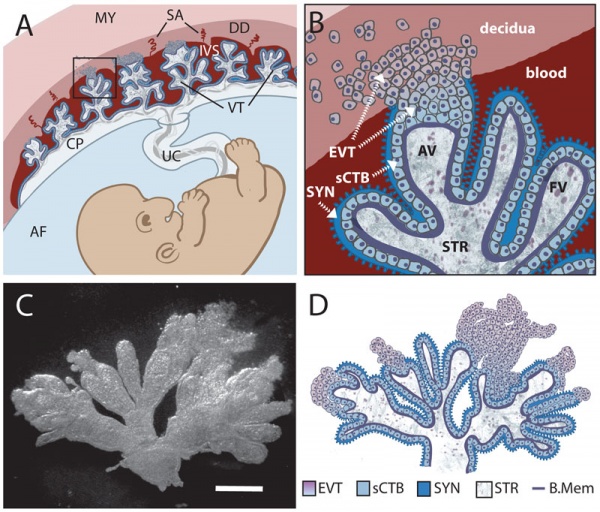
Cytotrophoblast Layer
There is a new interpretation of the changes that are occuring in the cytotrophoblast (CTB) layer during early to full-term human placenta development. Traditionally the interpretation was that the cytotrophoblast layer thinned and became discontinuous towards term. The thinning is thought due to the epithelium surface expanding at a faster rate than its volume. Two recent studies suggest that while the cytotrophoblast layer does indeed thin, it does not become discontinuous.
Syncytiotrophoblast Layer
The syncytiotrophoblast (STB) layer forms the epithelial covering of the entire villous tree. These cells are multinucleated, terminally-differentiated syncytium formed by the fusion of the underlying progenitor cytotrophoblast (CTB) cells. The process is described as "syncytialization" and is mediated by syncytin-1, an envelope protein of a human endogenous retrovirus W (HERV-W). The differentiation is regulated by chorionic gonadotropin (hCG) and the fusion of cytotrophoblast cells is ongoing during placental development.
Cellular parts derived from the syncytiotrophoblasts (apoptotic nuclei and microparticulate debris) can be shed into the maternal blood in which they are bathed. The apototic process appears to be part of the fusion mechanism between cytotrophoblast and the overlying multinucleate syncytiotrophoblast layer.
Studies have suggested that these cells are transcriptionally inactive. A recent study using a number of different detection techniques now suggests that at least some of the cells nuclei may still be transcriptionally inactive.