Paper - An Early Human Embryo (No. 1285, Manchester Collection), with Capsular Attachment of the Connecting Stalk
| Embryology - 19 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Florian J. Hill JP. An early human embryo (no. 1285, Manchester Collection), with capsular attachment of the connecting stalk. (1935) J. Anat., 69(4): 399-411. PMID 17104547
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
An Early Human Embryo (No. 1285, Manchester Collection), with Capsular Attachment of the Connecting Stalk
By J. Florian, M.D. and J. P. Hill, D.Sc., F.R.S.
From the Department of Anatomy and Embryology, University College ,London
- Links: Hill Collection
THE specimen described herein (No. 1285 of the collection of the Department of Anatomy, University of Manchester) was placed at our disposal by Prof. J. S. B. Stopford, F.R.S., and to him we desire to express our very grateful thanks for his generous action.
In our paper (1931) on the Dobbin embryo, we included, for comparative purposes, an account of the head-process and prochordal plate of this embryo and provided a graphic reconstruction of the dorsal view of the embryonal shield (1931, text-fig. 5), and although its state of preservation is not perfect, we think the chorionic vesicle as a whole is well deserving of brief description, since it exhibits certain features of unique interest.
History of the Specimen
Prof. Stopford informs us that he received the pregnant uterus (which provided the specimen) from his colleague, Prof. Daniel Dougal, M.C., M.D., some few hours after its removal. Unfortunately it is not now possible to obtain the full history of the case, but Prof.Dougal,although he did not himself perform the operation, has very kindly given us such information as he possesses concerning it. The uterus was removed by hysterectomy, following on chronic metritis. From the notes taken by the house-surgeon, pregnancy was not suspected, and the operation was undertaken by one of Prof. Dougal's colleagues. When the uterus was exposed, its appearance suggested to him the possibility of a nearly pregnancy. Some hours after the completion of the operation, Prof. Dougal had occasion to examine the uterus, and recognising its value, took it immediately to Prof. Stopford.
On looking more carefully into the record of the menstrual history of the patient,Prof. Dougal found that she had a normal periodicity of about 21 days and that the actual time intervening between the last menstrual period and the date of operation was 28 days, so that she had actually overrun a missed menstrual period by about a week.
If we assume that fertilization occurred at mid-interval, say on the 11th day of the cycle, and that the 28 days' interval is reckoned from the onset of menstruation, the fertilisation-age of the present specimen would be 17 days, which we think is too low, but the available data are not exact enough for an accurate determination.
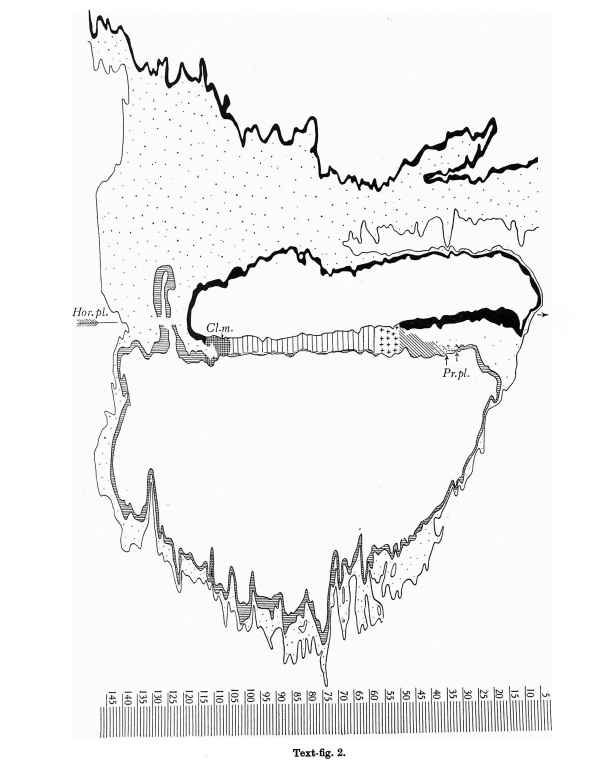
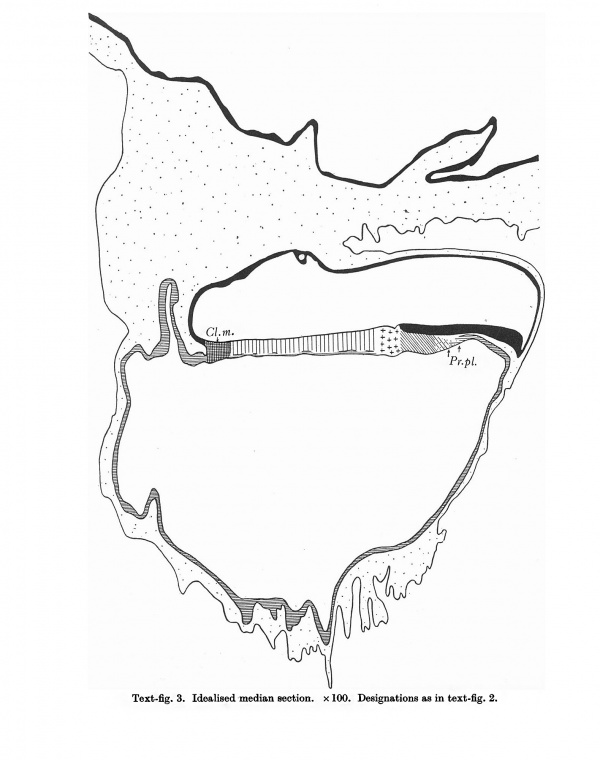
The specimen, comprising the completely implanted chorionic vesicle with the embryonal primordium and the immediately surrounding portion of the decidua, came into our hands as a very fine, complete series of sections which had been cut at a uniform thickness of 8P. We have numbered the sections containing the embryo and those immediately adjoining the latter in serial cranio-caudal order from 1 to 160.The embryo has been cut in a very favourable plane for study; the sections are nearly transverse to the embryonic axis (the difference between the sectional plane and the transverse plane being 90 30' as can be seen from our reconstruction of the dorsal view (text-fig.1))and are practically perpendicular to the plane of the embryonal shield. As our graphic reconstruction of the median section (text-fig. 2) shows, the outline of the embryo (especially that of the ventral wall of the yolk-sac and the chorionic ectoderm) is very uneven. This is due to variations in the dorso-ventral dimensions of the sections, the result of unequal expansion. The specimen, moreover, exhibits distinct signs of shrinkage of the embryonal vesicles, as the result of which the embryonal ectoderm has become largely detached from the underlying layers. This detachment is most marked in the cranial part of the embryonal shield and in the region of the cloacal membrane, the caudal part of which has become artificially separated into its constituent layers. Likewise the allantoic canal has been broken across and separated into two portions. In view of these defects, we have thought it advisable to provide, along with the accurate graphic reconstruction of the median section (text-fig. 2), an idealised figure of the same in which obvious imperfections have been corrected (text-fig.3).
Measurements
Chorionic vesicle including vili= implantation cavity: polar diameter 5mm.; maximum transverse diameter 4-4mm.
Chorionic cavity without vili=exocoelom: polar diameter 428mm.; transverse diameter 3-28 mm. (The foregoing measurements relate to section 160.)
Amniotic cavity: length 0 91 mm.; height 0-23 mm.
Embryonal shield, including cloacal membrane: length 0-87mm. (text- fig.2);breadth0-625mm. (text-fig.1).
Cloacal membrane: length 0.06mm.
Primitive streak: length 0.39 mm.
Hensen's knot: length 0.05 mm.
Head-process: length 0-125 mm.
Prochordal plate:length 0.03mm.
Yolk-sac cavity (text-fig. 2): Cr.-Cd. diameter (length) 0-98 mm., vertical diameter (height)074mm.,transverse diameter 0.988mm. (in section 80).
Allantoic canal:length (allowing for break),0.23mm.; thickness of bulbous upper end 0.04 mm.
The chorionic vesicle of pear-shaped outline in section (Plate III, fig. 11) is implanted in the decidua immediately to the left of a deep groove, the lower part of the decidua capsularis and the decidua marginal is actually forming the boundary of the groove on its side next the vesicle. From the figure, it will be seen that the embryo is attached to the chorion, not at the deep pole of the vesicle as is normally the case, but at its superficial pole, underlying the capsularis. This is unquestionably the most interesting feature displayed by the present specimen. It had already been noted by Prof. Stopford, and, so far as we are aware, the present specimen appears to be the only early human embryo in which this condition has actually been found to occur. In his invaluable book, Frihentwicklung, u.s.w., published in 1927, Grosser states (p.207) that: " ...junge Stadien mit ausgesprochen kapsularer Lagerung des Keimes, die vorkommen mfissen,wenn eseineprimer falschorientierte Implantation polar differenzierter Eiergibt, sindbisher nicht bekannt".
We refer below (p.407)to the significance of this condition.
The Embryo
(a) Amnio-embryonal vesicle
The embryo consists of amnio-embryonal and yolk-sac vesicles enveloped by the embryonal mesoderm, and is attached by the short but massive connecting stalk to the upper polar region of the chorion immediately below the capsularis. The amnio-embryonal vesicle is distinctly the smaller of the two. The embryonal shield forming its floor exhibits a definite Hensen's knot (PlateI,fig.1)situated a little in front of its centre,and extending forwards from the knot, to terminate in the prochordal plate thickening, is the short head-process(text-figs.1-3).We described the head-process and prochordal plate of this embryo in our paper of 1931 as follows:"So far as we can determine,the head-process is only very little longer than in [Florian's embryo] Bi24, but it is wider and thicker [Plate I, figs. 2-4]. It is accompanied, as in the latter, by lateral sheets of primitive streak mesoderm, which are much thicker than in Bi24. Owing to the state of preservation, the limits between the lateral mesoderm and the median head-process are somewhat indistinct in the caudal region. Cranially (text-fig.5) [our own text-fig.1], the mesodermal sheets become thinner,and the head-process, which is still thick and compact, is distinctly delimited from them. Still more cranially, at the level of the interrupted line in text-fig. 5 [our text-fig. 1], the head-process broadens out and the lateral sheets of mesoderm practically disappear. We regard the just mentioned line as marking the cranial limit reached by the primitive streak mesoderm accompanying the head-process. Cranially to that line the head-process narrows forward and is surrounded anteriorly and laterally by thickened endoderm, which we interpret as prochordal plate. If this explanation is right, the prochordal plate [Plate II, fig.5] appears to be much broader than that of Bi 24, but is of about the same length in the median plane and is in a little more advanced condition" (1931, pp. 467 and 468). WVe have nothing new to add to this description of the head-process and the prochordal plate. Behind Hensen's knot lies the well-marked primitive streak, here distinctly longer than in the next younger stage so far described, viz. embryo Bi 24 (Florian). At its caudal end, a distinct cloacal membrane is developed (text- figs. 1A3, Cl.rn.), its two constituent layers having become artificially separated in its caudal half.
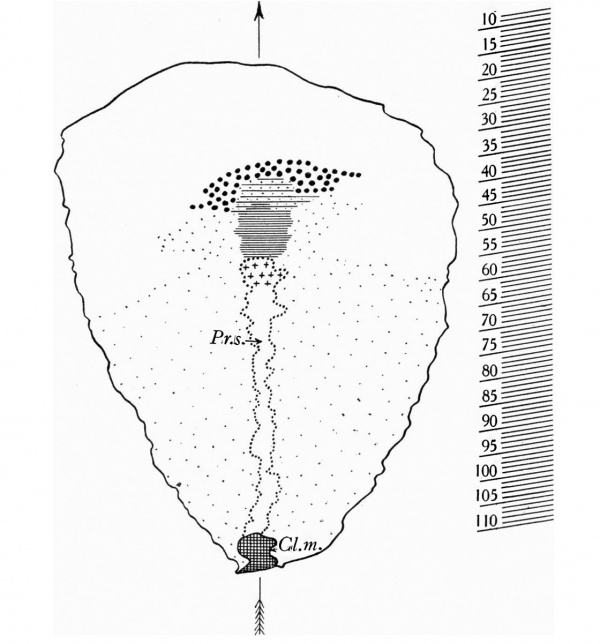
Text fig.1. Graphic reconstruction of the dorsal view of the embryonal shield. x100. The outline of the embryonal shield marked by a thick line. Outline of the primitive streak (Fr.s.) and Heunsen's knot marked by a dotted line; Heusen's knot marked by crosses. Cloacal membrane (C'l.m.) cross-hatched. Head-process marked by horizontal lines closely arranged in its compact, thicker caudal part (chorda process), more widely spaced with dots between, in its wider and more loosely constituted cranial part. prochordal plate marked by large dots. Secondary mesoderm below The embryonlal shield marked by by fine dots. On the right of the figure, levels of sections 8-110 are indicated. Median plane marked by an arrow.
In text-fig. 1 we have indicated by fine dots the area which we regard as occupied by mesoderm of primitive streak origin. Laterally to the primitive streak, the mesoderm would seem to reach the lateral margins of the embryonal area, whilst in front of Hensen's knot it extends forwards alongside the head- process as two lateral wing-like extensions. Even in well-preserved embryos, the limits between the primitive streak mesoderm and the primary mesoderm are difficult to determine, and in this specimen,the limits we have indicated in text-fig. 1 must be regarded as approximate only.
In front of the area in which we consider primitive streak mesoderm is present below the shield-ectoderm, we find the primary mesoderm occupying a zone situated around the cranio-lateral margin of the embryonal shield, whilst an area situated in front of, and laterally to, the prochordal plate is practically devoid of mesoderm.
The ectoderm of the embryonal shield passes at its lateral and cranial margins rather abruptly into the very flattened ectoderm of the amnion, and, at its caudal margin, into the stalk-ectoderm. The latter covers the cranio- proximal part of the umbilical stalk(seeFlorian,1930,p.457)and is composed of a layer of rather high cels with an irregular inner contour. Some of the cels seem to be in process of being shed into the amniotic cavity. In the caudal part of the latter is a quantity of cellular detritus (Plate II, fig. 8), very probably derived from these desquamated cells. A similar detritus has been found in the amniotic cavity in other embryos of about this stage of development. In connection with this phenomenon, we may direct attention to the occurrence in sections 77-83 of a small rounded piece of stalk-mesoderm, covered with flattened stalk-ectoderm which projects into the amniotic cavity and is evidently destined to be liberated into the same. Florian(1930)has described a comparable separation of stalk-mesoderm in his embryo BiXI (with ten paired somites). Here a much larger portion of stalk-mesoderm was found lying free in the amniotic cavity. According to Florian,this separation is effected by the activity of the stalk-ectoderm which penetrates into the stalk-mesoderm, and its object is to enable the amniotic cavity to expand at the expense of the tissue of the connecting stalk so as to provide room for the development of the tail- end of the embryo.
As can be seen in text-fig. 2, the amniotic mesoderm near the attachment of the amnion to the connecting stalk (between sections 66-53) is distinctly thicker than in the more cranial part of the amnion. In the sections it can be seen that between the mesodermal and ectodermal layers of the amnion there is present a thick layer composed of a network of fibrilae. We have here to do with a mesostroma in which the fibrilae,for the most part,run radially to the surface of the amnion, i.e. pass from the mesoderm to the ectoderm. Fahrenholz (1927) described this "thickening" under the name "Aufhingeapparat" ("suspensory apparatus"). It is developed in al specimens belonging to this developmental stage. In the sections, traces of the "suspensory apparatus" can be followed cranially to about section37. Stil more cranially,the mesoderm seems to be composed of a flattened layer of cells situated close to the flattened amniotic ectoderm.
(b) Yolk-sac
The structure of the ventral wall of the yolk-sac calls for brief mention. Whilst the endoderm of its cranial and lateral walls is formed of flattened cels, that of its ventral wall (beginning at about section 38 and caudally thereto) is distinctly higher. In this same region the mesodermal so becomes markedly thickened; it is composed of loose mesenchyme in which vesicular spaces of varying size are present,and it is produced out into irregular processes so that its surface-contour is extremely uneven (text-fig.2 and Plate I,fig.8,nt.).
This thickened region would seem to correspond with the site of origin of the yolk-sac process of other early embryos.
The thickening of the endoderm becomes specially marked over a medio-ventral area extending from about section 69 back to about section104. Here the endoderm is irregularly folded and provided with small diverticula (PlateI,fig.8,d.e.);its cels are columnar and contain numerous vacuoles, sometimes quite large, and also coarse spherules, often with smaller deeply staining granules inside them. These spherules appear to be shed into the yolk- sac cavity and to give rise to a coarse detritus,readily distinguishable from the very fine granules derived from the coagulated yolk-sac fluid.
Between sections 94-101 in this same region of the yolk-sac, a curious villous-like process composed of vacuolated cels projects freely from the endoderm into the yolk-sac cavity. Its stalk of attachment is so thin that it might easily become free in the latter.
Behind section 104, the columnar endoderm of the ventral wall gives place to cubical,and caudally to section 119, the latter passes over into the flattened epithelium of the caudal wall.
It remains to be mentioned that vessel-primordia are in process of differentiating in the mesoderm of especially the ventral and lateral parts of the yolk-sac wall.
Allantoic canal
Directly behind the cloacal membrane, a small area of the endoderm of the dorsal wall of the yolk-sac is definitely thickened and exhibits near its centre a minute funnel-shaped opening. This leads into a tubular diverticulum, the allantoic canal, which runs vertically upwards in the mesenchyme of the connecting stalk, immediately behind the caudal extremity of the amniotic cavity (text-figs.2 andaand PlateIII,fig.9). The separation of the canal into two parts in the sections is obviously an artifact. Its wall is formed of a layer of cubical endoderm which is somewhat thinner over the lower two-thirds of its cranial wall than elsewhere, with the result that the upper extremity of the canal appears bulbously thickened.
Text-fig.2. Graphic reconstruction of the median section. x100. Ectoderm black. Endoderm indicated by horizontal lines. Primary mesoderm dotted. Primitive streak marked by vertical lines. Cloacal membrane (Cl.m.) cross-hatched. Hensen's knot marked by crosses. Head-process marked by oblique lines closely arranged in its compact, caudal part, more widely spaced and with dots,in its wider and looser cranial part. Prochordal plate(Pr.pl.) marked by horizontal lines and dots. Horizontal plane(the plane of projection of text-fig.1) marked by an arrow (Hor.pl.). Levels of sections 2-147 indicated on the scale below the text-figure.
Text-fig.3. Idealised-median section. x100. Designations as in text-fig.2.
Connecting stalk
The connecting stalk of our specimen is comparatively short but is of considerable cranio-caudal extent, since its attachment to the amnion begins near the mid-region of the latter. It contains distinct vessel-primordia. In section 97 (Plate II, fig. 7), the primordia of the two umbilical arteries (u.a.1 and u.a.2) are distinctly visible. They can be traced nearly to the chorionic mem- brane. In this section, a narrow deep involution of the mesothelium (v.p.) of the connecting stalk suggests the participation of that layer in the formation of the umbilical vessels, as is believed to be the case in other early human embryos.
As we have already indicated, the most interesting feature exhibited by this embryo is provided by the attachment of the connecting stalk to the chorion. Whereas in other described early embryos this attachment lies adjacent either to the decidua basalis or to the marginalis, in the presentembryo it occurs below the decidua capsularis in a position diametrically opposite the basalis(PlateIII,fig.11,c.s.).
As in the normally attached embryo, the vili below the capsularis are much less developed than those in relation to the marginalis and basalis. Whilst the latter vili are elongated and exhibit distinct branching and are disposed more or les perpendicularly to the chorionic surface, those below the capsularis are smaller, less numerous, are much less branched and tend to be disposed tangentially to the chorion owing to the narrowness of the intervillous blood- space. From the area of the chorion to which the connecting stalk is attached a few small vili originate, but immediately to the left of the area of attachment isa small circumscribed region wholly devoid of vili.
The attachment of the connecting stalk to the upper or capsularis pole of the chorion where obviously the definitive placenta cannot develop, provides the explanation of the polar variety of the so-called insertio velamentosa of the umbilical cord, i.e. the condition in which the umbilical cord is attached to the chorion laeve opposite the placenta, and the umbilical vessels traversing that membrane enter and leave the organ all round its circumference. Thissame explanation has already been advanced by other workers, notably R. Meyer (1923), Schmidt (1925, p. 666), 0. Grosser (1927, p. 396), and is now, we believe, generally accepted,though,as we have already pointed out,the present specimen is the only early embryo in which the capsular attachment has been demonstrated.
Whilst, then, the velamentosous condition may be regarded as satisfactorily explained, how are we to account for the reversed implantation in the present specimen? We have no satisfactory explanation, beyond that of a chance happening. The deep-seated position of the embryonal primordium in the chorionic vesicle,adjacent to the decidua basalis, is generally ascribed to the coincidence of the upper pole with the implantation pole, i.e. to the fact that they are one and the same. In our specimen it would seem that this co-incidence for some reason failed and the lower pole functioned as the implantation pole. We are assuming that implantation is effected during the early blastocyst stage when the attachment of the inner cell-mass (embryoblast) clearly differentiates the poles and that normally implantation is a function of the trophoblast of the upper pole. On the other hand,if implantation takes place during the morula stage, and if the poles of the morula are already pre-determined, another possibility suggests itself to the effect that when the blastocyst cavity appeared, the inner cell mass adhered to the trophoblast of the lower pole instead of to that of the upper as is normal. In this case we should be dealing in our specimen with an apparent, not a real, reversal of the polarity of the chorionic vesicle.
Like Grosser (1927, p. 212), we are quite unable to accept the ideas put forward by v. Mollendorff (1921, 1925, 1926) to account for the position of the embryo at the deep pole of the chorionic vesicle. He holds that the embryoblast is originally intercalated in the trophoblast, and during implantation is the last part to enter the decidua.The embryonal primordium is thus originally situated below the capsularis, and he supposes it reaches its normal position in relation to the basalis either throught he rotation of the chorionic vesicleas a whole or by the active migration of the primordium by way of the chorionic mesoderm to its final site. In our opinion, if such a change in the position of the embryonal primordium occurs at al, which we very much doubt, it could only be effected by the unequal growth of the chorion and not in either of the ways envisaged by v. Mollendorff. In any case we hold that the original attachment of the embryoblast to the trophoblastal so marks the site of attachment of the later developed connecting stalk to the chorion, and in proof thereof we would point to the fact that the amniotic duct which we regard as owing its origin to that primary attachment, is always situated in the connecting stalk mesoderm.
Decidua
The decidua shows the typical characters of the premenstrual phase. The vesicle occupies nearly the whole thickness of the compacta, only a thin layer ofthe same underlying it.The epithelium of the glands exhibits the characteristic folding and the gland-lumina contain secretary coagulum and sometimes also extravasated blood. The decidual reaction is indicated in the decidua basalis and in the marginalis on the rightside next the groove as well as in the decidua parietalis on thesame side,but is litle evident in the decidua capsularis and in the marginalis on the left side. This latter region presents an oedematous appearance, and both it and the capsularis contain blood-extravasations. Large numbers of leucocytes a represent in the extravasated blood in the glands as well as in the decidual tissue,especially in the marginalis and the adjacent capsularis on the left side of the vesicle.
The capsularis is somewhat damaged and in the sections is partially separated into a poorly preserved outer zone containing much fibrinoid and a deep zone in rather better condition. Adjacent to the attachment of the connecting stalk, a thick flake-like portion of the outer zone,composed largely of fibrinoid, with only a few cells imbedded in it, is separated off in the sections. In places in the deep zone, traces of what Florian (1928) has termed the "Proliferations plasmodium" are recognizable. An epithelial layer is not distinguishable over the capsularis.
Decidual vesels. A number of small vessels of the nature of distended capillaries lead from the basalis to open into the intervillous blood spaceatthe deeppoleofthevesicle. In the interglandular tissue of the basalisitself, numbers of small arterioles and venules are present, but taken as a whole, the portion of the basalis available to us is entirely devoid of large vessels and cannot be described as richly vascularised. On the other hand,it is noteworthy that the only conspicuously large vessels in the immediate proximity of the vesicle are situated in the capsularis and in the marginalis on itsleftside.We regard these vessels as veins. They are enlarged and sinus-like, and their lumina are largely occupied by blood-corpuscles (Plate III, fig. 11, cv., cv.1). The largest of these veins is situated in the capsularis adjacent to the attachment of the connecting stalk (Plate III, fig. 11, cv.). It can be traced from behind the embryonal primordium forwards to its cranial extremity but is much reduced in diameter in the more cranial part of its extent. Inthecap- sularis, to the left of this main vein, there are present smaller veins (Plate III, fig.11,cv.1) which open into it as well as into the intervillous blood space on the upper leftside of the vesicle,whilst they are also connected with aw ell-marked vein which passes almost vertically downwards in the marginalis (Plate III, fig.11,mv.).
Chorion and villi
The chorion and vili exhibit the normal structure, the chorionic mesoderm being well developed. Round the periphery of the implantation cavity and between the extremities of the vili, there occur numerous irregular masses and strands of syncytio-trophoblast and also irregular disconnected masses of cytotrophoblast representing the remains of the basal trophoblast of earlier stages.The syncytio-trophoblast is most abundant on the surface of the basalis underlying the more cranial part of the vesicle. Plate III, fig. 10, from section 28, shows a canalised mass of it penetrating into a distended capillary in the basalis.
Summary
The specimen described was obtained from a case of total uterine extirpation and comprises the chorionic vesicle with the surrounding decidua and the contained embryo. The latter in its stage of development lies between Florian's embryo Bi24 and Grosser's embryo Wa 17. It is unique in that its connecting stalk is attached to the chorion not at its deep (basalis) pole as is normal but at its superficial pole underlying the capsularis. It thus provides the explanation of the polar variety of insertio velamentosa of the umbilical cord.
References
FAHRENHOLZ, C. (1927). Z. mikr.-anat. Forsch. Bd. vmy. FLORAN, J. (1928). Anat. Anz. Bd. LXVI, Erg.-Heft.
- (1930).J.Anat., Lond.,vol.LXIV.
GROSSER,0. (1927). Frilhentwicklung, Eihautbildungu. Placentation des Menschen der Sdugetiere. Miinchen: J.F. Bergmann .
HILL,J.P.andFLORIAN,J.(1931). Philos.Trans. B,v ol.ccxix.
MEYER, R. (1923). Arch. Gynak. Bd. cxvi.
V. M6LLENDORFF, W. (1921). Z. ge8. Anat. 1.Z. Anat. EntwGeach. Bd. LXII.
- (1925). Z.ge8.Anat.1.Z.Anat. Enti Geach. Bd. LXXVI. (1926). Z.mikr.-anat.Forach.Bd.v.
SCHMIDT, H.R.(1925). "Pathologie der Decidua, der Eihauteu. der Nabelschnur" in Lief.18 of Halben, J. and Seitz, L., Biologie. Pathologie des Weibes. Wien: Urban und Schwarzenberg.
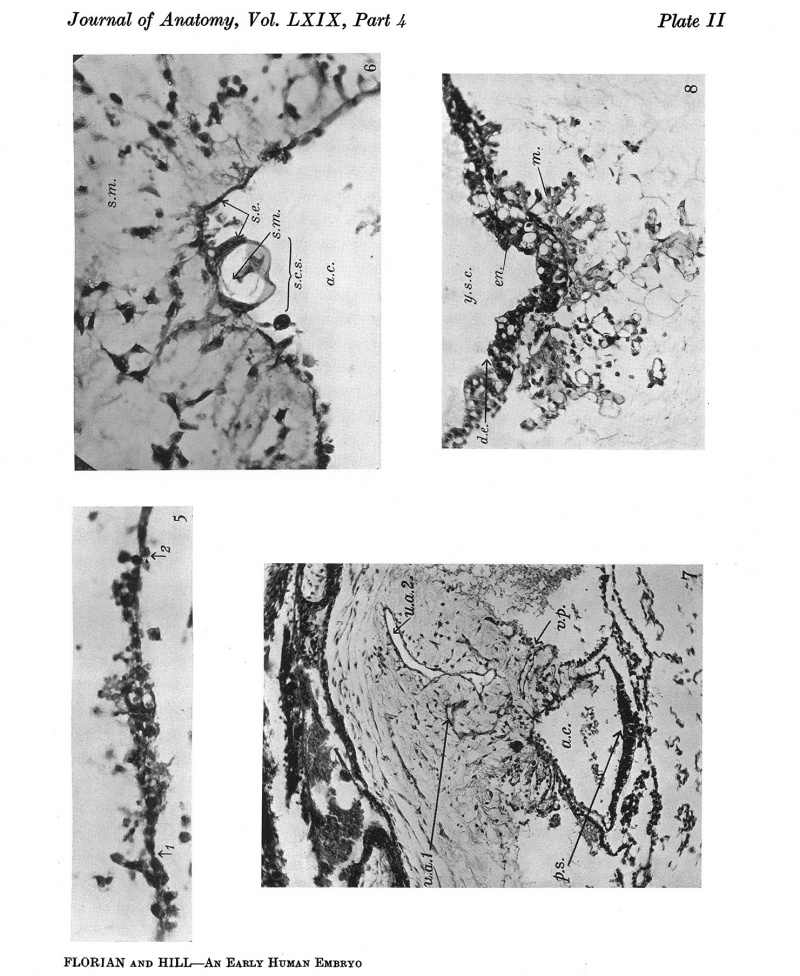
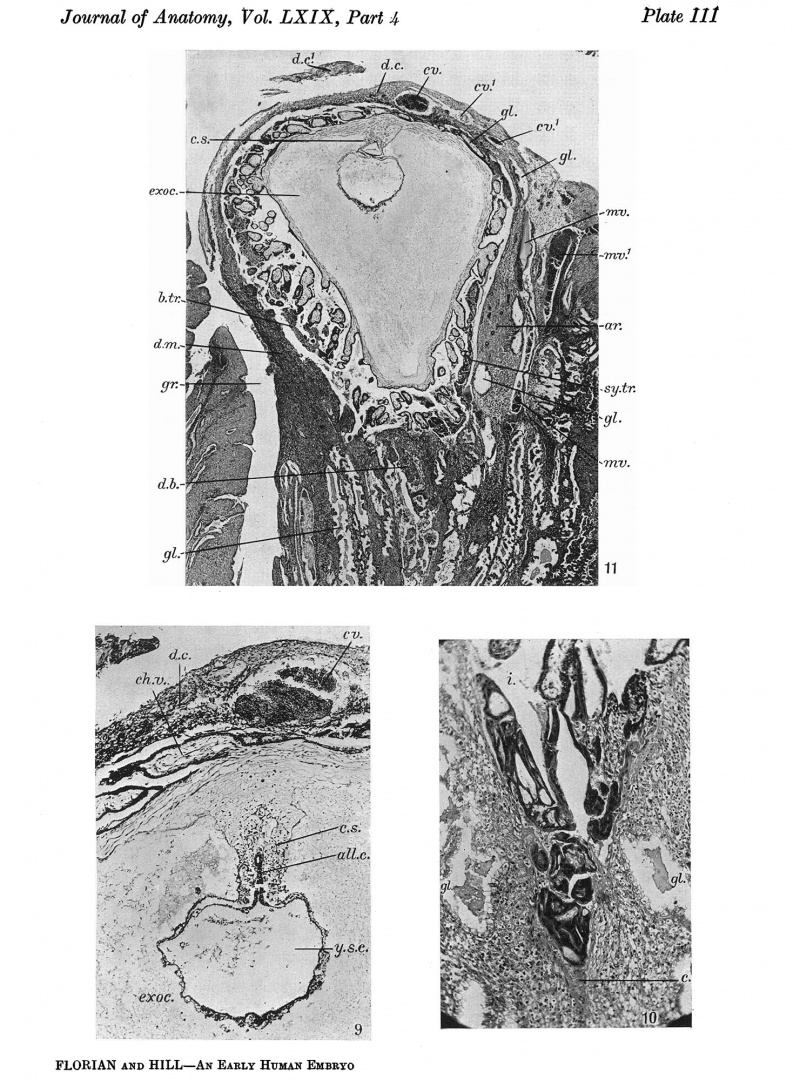
Explanation of Plates I-III
Plate I
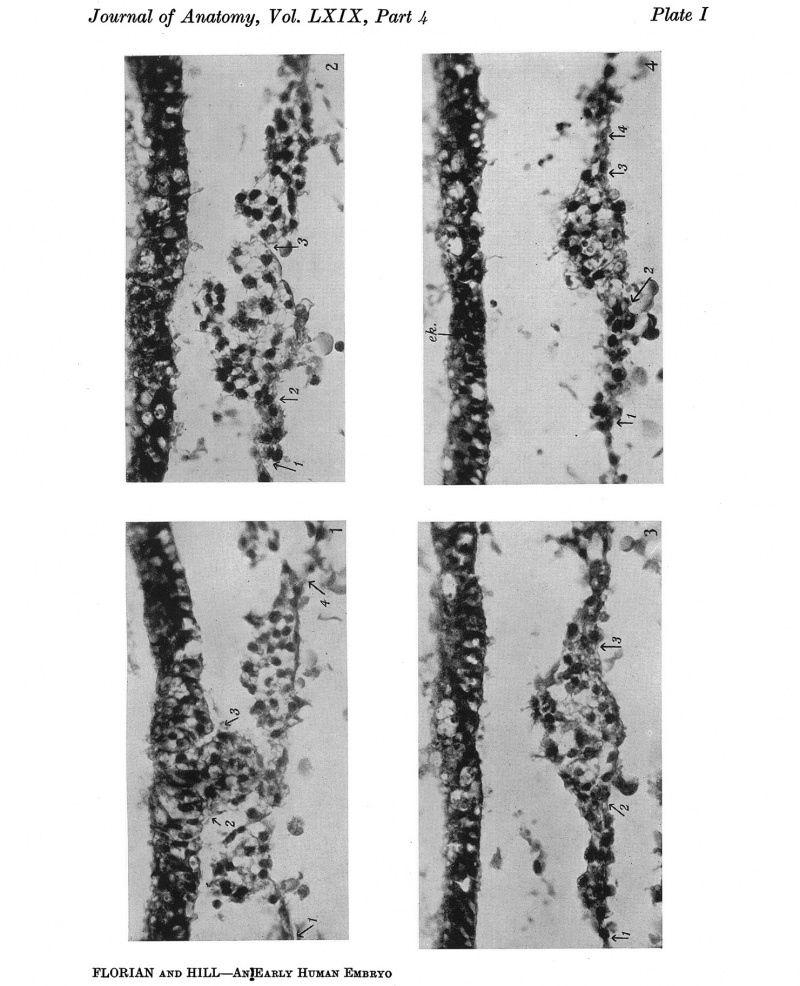
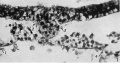
Fig. 1. Section 49, passing through the cranial extremity of Hensen's knot, which is situated between arrows 2 and 3. The lateral margins of the primitive streak mesoderm are indicated by arrows 1 and 4. x391.
Fig.2. Section 43, passing through the caudal, more compact part of the head-process (chorda-process), situated between arrows 2 and 3. The lateral margin of primitive streak mesoderm indicated by arrow 1; the margin on the other side is not included in the figure. x391.
Fig.3. Section 40, passing through the more cranial part of the chorda-process. Designations as in fig.2. x391.
Fig.4. Section 37, passing through the wider, less compact cranial part of the head-process (between arrows 2 and 3). The lateral margins of primitive streak mesoderm indicated by arrows 1 and 4. ek., ectoderm of the embryonal shield, artificially detached from the underlying tissue. x391.
Plate II
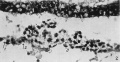
Fig.5. Section 32, passing through the prochordal plate and the cranial extremity of the thin part of the head-process (between arrows 1 and 2). x391.
Fig. 6. Section 79, passing through the sequestered mesoderm of the connecting stalk (above "s.C.s."). a.c. amniotic cavity. s.e. stalk-ectoderm. a.m. stalk-mesoderm. Dorsal side above. x391.
Fig.7. Section 97, passing through the connecting stalk and the primitive streak. In the connecting stalk, the primordia of the umbilical arteries (u.a.1andu.a.2)canbeseen.The mesothelium of the connecting stalk shows a deep invagination at v.p.(vessel-primordium). a.c.am- nioticcavity. p.s.primitivestreak. x115.
Fig.8. Section 75, passing through the ventral wall of the yolk-sac(see text, p.404). y.s.c.=yolk- sac cavity. en. endoderm. d.e. diverticulum of the endoderm directed towards the mesoderm. m. mesoderm. x186.
Plate II I
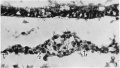
Fig.9. Section 122, passing through the connecting stalk(c.s.)andtheallantoiccanal(al.c.). ch.v. chorionic villus. cv. vein in decidua capsularis (d.c.). exoc. exocoelom. y.s.c. yolk-sac cavity. x48.
Fig. 10. Section 23 A large mass of syncytio-trophoblast is invading a distended capillary (c.). i.intervillous space. gl. uterine gland. x104.
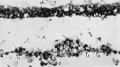
Fig.11. Section 99 General sectional view of the chorionic vesicle and the surrounding decidua. The section (99) passes through the embryonal primordium at the level of the hinder end of the primitive streak and shows the amnio-embryonal vesicle, here much reduced in size, the very much larger yolk-sac vesicle and especially the connecting stalk attached to the chorion below the central region of the decidua capsularis. ar. arteriole in d. marginalis. b.tr. basal trophoblast. c.s. connecting stalk. cv., cv.1 veins in d. capsularis (d.c.). d.c.' portion of capsularis artificially separated. d.b. decidua basalis. d.m. d. marginalis. exoc. exocoelom. gi.uterine gland. gr. groove in decidua.mv.,mv.' veins in d.marginalis. sy.tr. syncytio-trophoblast. x16.
Reference
Florian J. Hill JP. An early human embryo (no. 1285, Manchester Collection), with capsular attachment of the connecting stalk. (1935) J. Anat., 69(4): 399-411. PMID 17104547
Cite this page: Hill, M.A. (2024, April 19) Embryology Paper - An Early Human Embryo (No. 1285, Manchester Collection), with Capsular Attachment of the Connecting Stalk. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_An_Early_Human_Embryo_(No._1285,_Manchester_Collection),_with_Capsular_Attachment_of_the_Connecting_Stalk
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G