File:Protein-protein interaction.jpg
Original file (600 × 776 pixels, file size: 85 KB, MIME type: image/jpeg)
Protein-Protein interaction in fertilisation
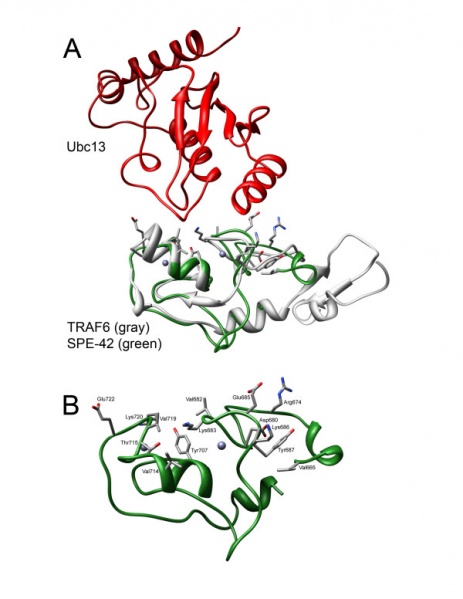
Putative protein-protein contact residues in SPE-42 homology model.
(A) Interaction between TRAF6 (gray) and Ubc13 (red) including the SPE-42 RING domain (green) aligned for comparison. TRAF-6 was co-crystalized with Ubc13 in pdb:3HCT. Following structural alignment of SPE-42, all residues within 5Å of Ubc13 were determined and shown. (B) SPE-42 RING domain alone with putative protein-protein interacting residues on SPE-42 surface shown. Individual amino acids are labelled and numbered in accordance with the complete SPE-42 amino acid sequence.
Reference
<pubmed>21345212</pubmed>
© 2011 Wilson et al; licensee BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Note - This image was originally uploaded as part of an undergraduate science student project and may contain inaccuracies in either description or acknowledgements. Students have been advised in writing concerning the reuse of content and may accidentally have misunderstood the original terms of use. If image reuse on this non-commercial educational site infringes your existing copyright, please contact the site editor for immediate removal.
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 18:51, 7 August 2012 |  | 600 × 776 (85 KB) | Z3331264 (talk | contribs) | ==Protein-Protein interaction in fertilisation== Putative protein-protein contact residues in SPE-42 homology model. (A) Interaction between TRAF6 (gray) and Ubc13 (red) including the SPE-42 RING domain (green) aligned for comparison. TRAF-6 was co-cry |
You cannot overwrite this file.
File usage
The following page uses this file: