2011 Group Project 11: Difference between revisions
No edit summary |
No edit summary |
||
| Line 92: | Line 92: | ||
The neural crest in each neromeric level is responsible for supplying it's specified zones of ectoderm and mesoderm; corresponding proteins are also expressed by the neural crest cells that complement the proteins secreted by the neural tube. The unique genetic markers associated with each neuromere can assist to identify the origins of craniofacial tissues that are eventually developed.<ref>Hunt P, Krumlauf R. Deciphering the Hox code: clues to patterning brachial regions of the head. Cell. 1991;66:1075–8. [PubMed]</ref> | The neural crest in each neromeric level is responsible for supplying it's specified zones of ectoderm and mesoderm; corresponding proteins are also expressed by the neural crest cells that complement the proteins secreted by the neural tube. The unique genetic markers associated with each neuromere can assist to identify the origins of craniofacial tissues that are eventually developed.<ref>Hunt P, Krumlauf R. Deciphering the Hox code: clues to patterning brachial regions of the head. Cell. 1991;66:1075–8. [PubMed]</ref> | ||
The neuromeric model facilitates a precise mapping of the anatomical site of origin for the zones of ectoderm and endoderm supplied by a particular area of the nervous system. The density of neural crest cell population in these areas also can help us understand their individual roles in generating the structures they do.<ref> Indian J Plast Surg. 2009 October; 42(Suppl): S19–S34.doi: 10.4103/0970-0358.57184 PMCID: PMC2825068 </ref> | |||
=Neural crest cells forming craniofacial structures= | |||
{|border="1px" cellspacing="1" align="center" cellpadding="2" | |||
! Neural Crest Cell Zone !! Structure Generated | |||
|- | |||
|'''Premaxilla and Vomer''' | |||
|The rostral aspect of the second rhombomere (r2)<ref>Indian J Plast Surg. 2009 October; 42(Suppl): S19–S34.doi: 10.4103/0970-0358.57184 PMCID: PMC2825068</ref> | |||
|- | |||
|'''The inferior turbinate, palatine bone, alisphenoid, maxilla and zygoma''' | |||
|The caudal aspect of the neural crest of rhombomere (r2))<ref>Indian J Plast Surg. 2009 October; 42(Suppl): S19–S34.doi: 10.4103/0970-0358.57184 PMCID: PMC2825068</ref> | |||
|- | |||
|'''The squamous temporal, mandible, malleus and incus ''' | |||
|The third rhombomere (r3)<ref>Indian J Plast Surg. 2009 October; 42(Suppl): S19–S34.doi: 10.4103/0970-0358.57184 PMCID: PMC2825068</ref> | |||
|- | |||
=Non neural crest cells forming craniofacial structures= | |||
{|border="1px" cellspacing="1" align="center" cellpadding="2" | |||
! Non Neural Crest Cell Zone !! Structure Generated | |||
|- | |||
|'''Cranial Base''' | |||
|PAM from somitomere 1<ref>Depew MJ, Simpson CA, Morasso M, Rubenstein JL. Reassessing the Dlx code: Genetic regulation of branchial arch skeletal pattern and development. J Anat. 2007;27:501–61.</ref> | |||
|- | |||
|'''Parietal Bone ''' | |||
|Epaxial PAM from somitomeres 2 and 3 <ref>Nowicki JL, Burke AC. Testing Hox genes by surgical manipulation. Dev Biol. 1999. pp. 210–238</ref> | |||
==Diagnosis== | ==Diagnosis== | ||
Revision as of 19:29, 14 September 2011
| Note - This page is an undergraduate science embryology student group project 2011. |
Cleft Palate and Lip
--Mark Hill 12:11, 8 September 2011 (EST) This project shows extremely poor progress at this time. May empty sub-sections and many of you have not met the requirements as required in an earlier laboratory assessment to add your content to the project page. Given the scope of your project topic there are an enormous number of resources, which have not been researched or utilised.
- Timeline would look better as bullet points with the date in bold as the first text.
- Only figure added is incorrectly named File:Pierre-Joseph_Desault-jpg.jpg. I had previously contacted and asked for this to be fixed and a citation added, neither has been done.
- I cannot provide you any further constructive comments at this stage, because there is insufficient for me to work with.
Introduction
--Mark Hill 12:10, 8 September 2011 (EST) There is no introductory text here.
- Cleft lip more prevalent in males
- Cleft palate more prevalent in females; 60:40 ratio females: males
- Neither age nor parity appear to be directly related
- Frequency of this birth defect is about 1 in 2,000 newborn in white populations
- It is higher in the Japanese
- lower in the Negro populations
History
In ancient times many congenital deformities, including the cleft lip and palate, were considered to be evidence of the existence of an evil spirit in the affected child. The reaction of the birth of a deformed child has varied widely from culture to culture where the infant was often removed from the tribe or cultural unit and left to die in the surrounding wilderness, a practise that was common in Antiquity and still happens today in certain parts of African tribes. In Sparta the unfortunate newborns were abandoned on Mount Tagete, while in Rome they were drowned in the Tarpeian rock.
The renowned philosopher Plato discussed it in one of his dialogues in the Republic, explaining that it was indeed a means of eradicating evil omens and preserving the soundness of the race.[1]
This state of lack of knowledge is evident up until 1889 when Keating published his opinion that a series of congenital anomalies were provoked in each case, by the mother looking at a person with similar deformity during her pregnancy.[2] The first persuasive explanation regarding the actual causation of the condition offered by Philippe Frederick Blandin between 1838 to1896 has revolutionised the perception of the condition. It sparked interest among physicians to investigate further into embryological development and the possible origin of clefting.[3]
Since 1896 to the 19th century both understanding and the surgery of cleft lip witnessed remarkable improvements. Surgeons around the world continued to research and propose refinements on the early procedure striving to accomplish precise and reproducible methods.
Timeline
- In 1295- 1351, Jean Yperman noted that cleft had a congenital origin classifying the various forms of the condition and outlining corresponding treatment principles. [4]
- In 1460, Heinrich von Pfolsprundt passed stitches through all the layers to repair the cleft instead of simply suturing the skin accomplishing a better repair of the lip. [5]
- In 1537-1619, Fabricius ab Aquapendente first described the embryological basis of cleft lip. [6]
- In 1561, Pierre Franco and Ambroise Pare described the techniques of correction of both unilateral and bilateral cleft lips in Traite des Hernies using dry sutures, pins and a triangular bandage. He emphasized that an accurate surgery procedure can produce an inconspicuous scar, an outcome which was “particularly desirable when the patient was a girl”.[7]
- In 1795, Pierre Joseph DeSault, a French pioneer of bilateral cleft surgery at La Charité and Hôtel-Dieu in Paris developed a new method for teaching anatomy and taught the procedure of bilateral cleft surgery[8]
- In 1808, Meckel pubished his theory of the embryological development of the lips which stated that the lips formed from five distinct processes which eventually united, three for the upper lip and two for the lower lip. [9]
- In 1838, Philippe Frederick Blandin suggested that facial cleft resulted from a failure of the premaxilla and the maxillary segments to unite at a later stage in development. [10]
- In 1844, Germanicus Mirault introduced a triangular flap from the lateral side into a gap created by making a horizontal incision on the medial side of the lip creating a nostril floor and reducing the linear scar on the lip. [11]
- In 1872, Jacob August Estlander, a Finnish surgeon introduced a method to correct the mid-face retrusion that was left by the bilateral lip repair process. He recommended a wedge resection of the vomer which allowed the protruding premaxilla to be pushed back. [12]
- In 1935, Faltin, another Finnish surgeon recommended that the procedure described by Jacob A Estander be abandoned because it routinely left serious maxillary retrusion. [13]
- In 1960, Peter Randall standardized the triangular flap repair method with accurate and reproducible measurements. [14]
- In 1965, W. M. Manchester introduced a procedure for the bilateral cleft surgery. [15]
- In 2000, Hua Xi Kou Qiang demonstrated that simultaneous primary palate repair and alveolar bone grafting are safe for unoperated cleft palate patients, and this procedure should be performed in unoperated cleft palate patients above 8 years old. [16]
- In 2008, J Y Wong published a study describing that craniofacial anthropometry using the 3dMDface System is applicable and reliable. Application of software algorithms merging the different overlapping images into a single three-dimensional image can remarkably improve landmark identification. [17]
- In 2010, B Mishra along with a team of Indian doctors published a study concluding that Nasoalveolar molding can be a useful adjunct for management of cleft lip nasal deformity. It serves as a cost-effective technique that can diminish the number of future surgeries such as alveolar bone grafting and secondary rhinoplasties in under developed countries. [18]
Neuroembryology and functional anatomy of craniofacial clefts
Embryological anatomy is the foundation upon which all treatment methods for craniofacial anomalies such as cleft must be based. Craniofacial clefts characterize states of inappropriate(excess or deficiency)distribution within and between specific developmental fields. The neuromeric mapping of the embryo is the primary denominator for understanding normal anatomy and pathology of the head and neck. [19]
The central nervous system of the human embryo develops in discrete segmental craniocaudal units called neuromeres. Specific genes known as Hox genes define the anatomical boundaries of each neuromere. [20]
The neural crest in each neromeric level is responsible for supplying it's specified zones of ectoderm and mesoderm; corresponding proteins are also expressed by the neural crest cells that complement the proteins secreted by the neural tube. The unique genetic markers associated with each neuromere can assist to identify the origins of craniofacial tissues that are eventually developed.[21]
The neuromeric model facilitates a precise mapping of the anatomical site of origin for the zones of ectoderm and endoderm supplied by a particular area of the nervous system. The density of neural crest cell population in these areas also can help us understand their individual roles in generating the structures they do.[22]
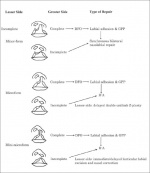
Neural crest cells forming craniofacial structures
Non neural crest cells forming craniofacial structures
| Neural Crest Cell Zone | Structure Generated |
|---|---|
| Premaxilla and Vomer | The rostral aspect of the second rhombomere (r2)[23] |
| The inferior turbinate, palatine bone, alisphenoid, maxilla and zygoma | The caudal aspect of the neural crest of rhombomere (r2))[24] |
| The squamous temporal, mandible, malleus and incus | The third rhombomere (r3)[25] |
| Non Neural Crest Cell Zone | Structure Generated | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cranial Base | PAM from somitomere 1[26] | ||||||||||||||||||||||||||||||||||||||||
| Parietal Bone | Epaxial PAM from somitomeres 2 and 3 [27]
DiagnosisCleft lip and palate occurrence varies among continents, races and populations from one in 700 to one in 1500 newborns. [28][29] CLP is implicated to be related to more than 100 syndromes, and trisomy 13 is the most commonly associated chromosomal anomaly. Associated malformations occur most commonly in the facial region (21%), followed by the ocular system, central nervous system, skeletal system, cardiovascular system, neck, auricular system, gastrointestinal system and urogenital system[30] While the birth of a child with CLP can have a severe emotional impact on the parents, antenatal diagnosis with ultrasonography helps to reduce the impact and prepare the parents psychologically. Most parents that received prenatal counselling following the diagnosis of CLP antenatally (85%) felt that the information had prepared them psychologically for the birth of their child, and 92% indicated that they had never considered the possibility of voluntary termination of pregnancy. Prenatal diagnosis of CLP has the additional advantage of allowing plastic surgeons to achieve early lip repair.[31] Prenatal ultrasound detection of the fetal face remains a matter of clinical research due to several technical difficulties associated with the examination of the fetus.[32] The most exigent obstacles faced during CLP examination are as follows:
Cleft Soft Palate DetectionThese technical challenges imply that the opportunity of prenatal screening for isolated defects of the soft palate is remote at present. Considering the additional time and effort involved in achieving satisfactory insonation and visualization, these methods are usually restricted to certain cases in which a cleft lip or hard palate is suspected. Isolated defects in the soft palate are relatively easier to repair surgically, and their emotional trauma on the parents if they are not diagnosed antenatally is far less severe than in newborns with cleft lip or cleft hard palate.[35] Cleft Hard Palate DetectionA combination of three different methods of 3D ultrasounds has been tested to measure detection accuracy of hard palate anomalies (See Table 1 and 2). The three methods are:
To date, none of the three methods (reverse face, flipped face or oblique face) have been found to be notably superior to the others. Therefore a combination of all three tests should be carried out for any fetus provided that there are adequate volume acquisition and reasonable time to examine the images offline. However, in cases where an immediate diagnosis is required, the fastest method is the oblique-face approach. On the other hand when initial volume acquisition is inadequate and image quality is less than optimum, the reverse-face view or the oblique-face view in coronal planes is the most effective technique to use. [36] Accurate ultrasound imaging of the secondary palate also requires the presence of fluid between the fetal tongue and palate, as well as curving of the plane so that the acquired volume follows the concave structure of the palate with either the oblique-face or flipped-face view. In some cases these views can reveal useful details about the soft palate which can be potentially be used in prenatal diagnosis of defects in future cases.
Table 1. Percentage of fetuses with cleft lip and palate (n = 10) in which abnormal findings were well visualized using each technique [37]
Table 2. Percentage of fetuses with cleft lip and palate (n = 50) in which abnormal findings were well visualized using each technique [38] Development of Disease--Mark Hill 12:18, 8 September 2011 (EST) There is no text here. Aetiology--Mark Hill 12:18, 8 September 2011 (EST) There is no text here.
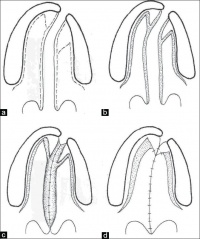
Developmental StagingDevelopment of the lip occurs during face development between weeks four and five. During this time the components that contribute to the face morphology come together and are fused to form a complete upper lip. These components include the maxillary processes and the medial and lateral nasal processes [39] At stage 15 the medial and nasal processes have started to fuse and the maxillary process lie inferior to them. During stage 16 the maxillary process and the medial nasal processes come in contact with each other and begin to fuse. Stage 18 is the later stage of lip formation. The maxillary process continue to grow and in doing so the force the medial nasal processes medialfrontally. It is between Stages 16 and 18 that a cleft lip is formed, after failure of the processes to fuse completely [39] Development of the palate occurs between the seventh and tenth weeks. There are two main stages in palate development, primary and secondary. During the seventh week of development the intermaxillary processes are formed. These processes give rise to the primary palate. This primary palate contributes to the floor of the nasal cavity. Towards the end of the seventh week the palatine shelves, which were lying parallel to the tongue, start to move into a more horizontal position above the tongue. These palatine shelves begin to fuse with each other and with the primary palate. Fusion is complete by week ten and the secondary palate is formed. It is between weeks seven and ten of development that a cleft palate is formed as a cleft palate is the result of the palatine shelves failing to fuse properly. [40] Types of Cleft Palate/LipThere are different types of both cleft palates and cleft lips, each with varying degrees of severity. Cleft palates and lips may either occur together or individually. Variations of cleft palate/lips include:
A cleft palate may be either complete or incomplete. It may also be unilateral or bilateral, and involve just the soft palate or include the hard palate as well. There are many variations of a cleft lip. Cleft lips may occur unilaterally or bilaterally. A unilateral cleft lip presents with only one cleft, either complete or incomplete. As with a cleft palate, a cleft lip may also be incomplete or complete. The bilateral cleft lip may also be further divided into Binderoid bilateral complete cleft lip and palate, Bilateral complete cleft lip and intact secondary palate, Bilateral incomplete cleft lip and Asymmetrical bilateral (complete/incomplete) cleft lip [42] Lesser forms of incomplete bilateral cleft lips can be divided into micro-form, microform, and mini-microform depending on the degree of the disruption at the vermilion-cutaneous junction [42] PathophysiologyThere is no text here. Genetic ConfigurationThere is no text here. Treatment
1. Hard palate repair techniques Veau-Wardill-Kilner pushback (V-Y) von Langenbeck bipedicle flap (W) Bardach’s two-flap (V) Alveolar extension palatoplasty (AEP) Vomer flap Raw area free palatoplasty 2. Soft palate repair techniques Intravelar veloplasty Furlow double opposing Z-plasty Radical muscle dissection Primary pharyngeal flap Two stage palatal repair 3. Protocol based techniques Schweckendiek's Malek's Hole in one Complications include: -Haemorrhage -Respiratory obstruction -Hanging Palate -Dehiscence of the repair -Oronasal fistula formation -Bifid uvula -Velopharyngeal Incompetence -Abnormal speech -Maxillary hypoplasia -Dental malpositioning and malalignment -Otitis media Current and Future ResearchThere is no text here. External LinksThere is no text here. Glossary/TermsThere is no text here. Gallery
File:Cleft lip 3.jpg Image shows Pre-Op Bilateral Cleft Lip
References
Textbooks2011 Projects: Turner Syndrome | DiGeorge Syndrome | Klinefelter's Syndrome | Huntington's Disease | Fragile X Syndrome | Tetralogy of Fallot | Angelman Syndrome | Friedreich's Ataxia | Williams-Beuren Syndrome | Duchenne Muscular Dystrolphy | Cleft Palate and Lip |